Significance
Graphene and Quinone or its derivatives composites present potential alternative organic molecules. Quinones are well-known as electrochemically active organic molecules and have been applied as energy storage materials for supercapacitors. Adoption of such redox active materials, that particularly do not include any minor metals resources for positive electrode materials as substitutes for the currently predominant lithium-based electrode in rechargeable batteries would help mitigate the imminent resource problem. To date, many types of potential organic materials have been studied where low molecular weight organic derivatives have become the epicenter of much research in recent years. 9,10-Anthraquinone (AQ) is an example which shows a high utilization ratio during the first discharge process; however, the electrode using AQ itself significantly deteriorates during cycling. This defect limits full adoption of AQ. Recent publications have pointed out that through the application of some special binder separator and electrolyte solutions containing additives, the defect has been partially overcome. Consequently, more research is needed.
To this end, in order to improve the cycling stability of AQ, suppression of the dissolution remains inevitable. In this view, a group of researchers from the National Institute of Advanced Industrial Science and Technology (AIST), Osaka, Japan: Dr. Masaru Yao, Dr. Hikaru Sano, Dr. Hisanori Ando, Dr. Tetsu Kiyobayashi and Dr. Nobuhiko Takeichi, proposed a novel approach focused on the oligomerization technique of AQ. To realize their goal, the scholars synthesized a novel AQ-dimer and trimer connected by the triple bond as positive electrode active materials following which they assessed the materials’ charge/discharge behavior in comparison to that of the AQ-monomer. Their work is currently published in the research journal, ChemPhysChem.
Specifically, their approach involved the synthesis of the aforementioned AQ-dimer and trimer connected by the acetylene unit by a palladium-catalyzed cross-coupling reaction. The researchers experimentally demonstrated the oligomerization process, i.e. connection of only a couple of redox-active organic units in a bid to extend the cycle life of the organic electrode without impairing the utilization ratio of the active units. In fact, they confirmed this process by comparing the anthraquinone monomer with its newly synthesized dimer and trimer. Lastly, quantum chemistry calculations were used to corroborate their experiments.
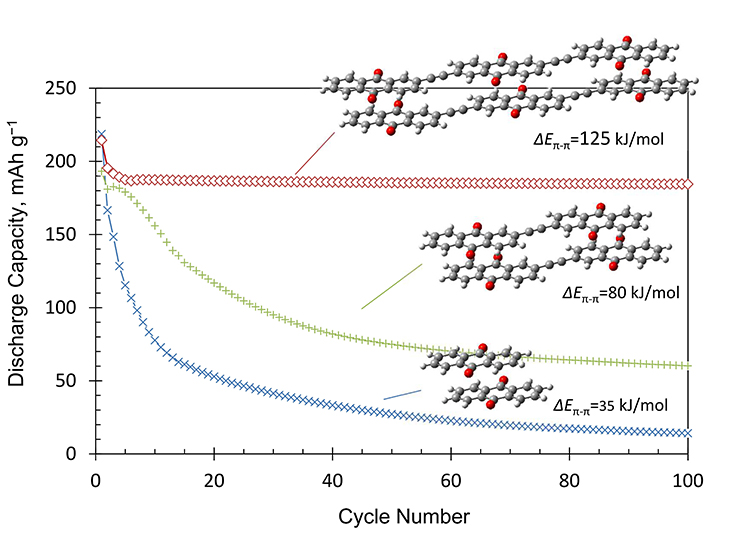
The authors reported that even though the capacity of the AQ-monomer quickly decayed upon cycling, the synthesized oligomers showed improved behaviors. In particular, the trimer showed the best cycling performance; it retained an almost constant capacity during the one hundred cycles. Further, the quantum chemistry calculations indicated that the attractive intermolecular π–π interactions in the oligomers were strong enough to stabilize the molecules in the crystal.
In summary, a dimer and trimer with the anthraquinone (AQ) skeleton connected by a triple bond (C≡C) were newly synthesized and their battery performance was compared to that of the AQ monomer. Remarkably, the results presented indicated that the oligomerization of the soluble redox active molecular units was effective to extend the cycle-life without lowering the discharge voltage. A theoretical quantum chemistry calculation also showed that the intermolecular attractive interaction through the π–π interaction could become high enough by the oligomerization to stabilize the molecule in the crystal, which should contribute to suppressing the dissolution. Overall, in an interview with Advances in Engineering, Dr. Masaru Yao – the lead researcher, pointed out that the findings of their study would offer guide to design a novel organic compound showing a high capacity and good cycling performance.
Reference
Masaru Yao, Hikaru Sano, Hisanori Ando, Tetsu Kiyobayashi, Nobuhiko Takeichi. Anthraquinone-Based Oligomer as a Long Cycle-Life Organic Electrode Material for Use in Rechargeable Batteries. ChemPhysChem 2019, volume 20, page 967 –971.
Go To ChemPhysChem Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.