Significance
Hydrogen has great potential as an alternative energy carrier due to its trifold higher gravimetric energy density when compared to current petroleum based fuels. Unfortunately, its efficient storage still poses critical issues which must be resolved before establishing its self-driven infrastructure. In the recent past, the quest for a suitable material capable of storing hydrogen safely has proven fruitful as materials such as amides, silanides and alanates have been found to be suitable. Conversely, issues relating to their thermodynamics or kinetics need to be resolved first before they can be considered ideal for hydrogen storage. Alternatively, magnesium and its complex variants have emerged as promising materials for hydrogen storage.
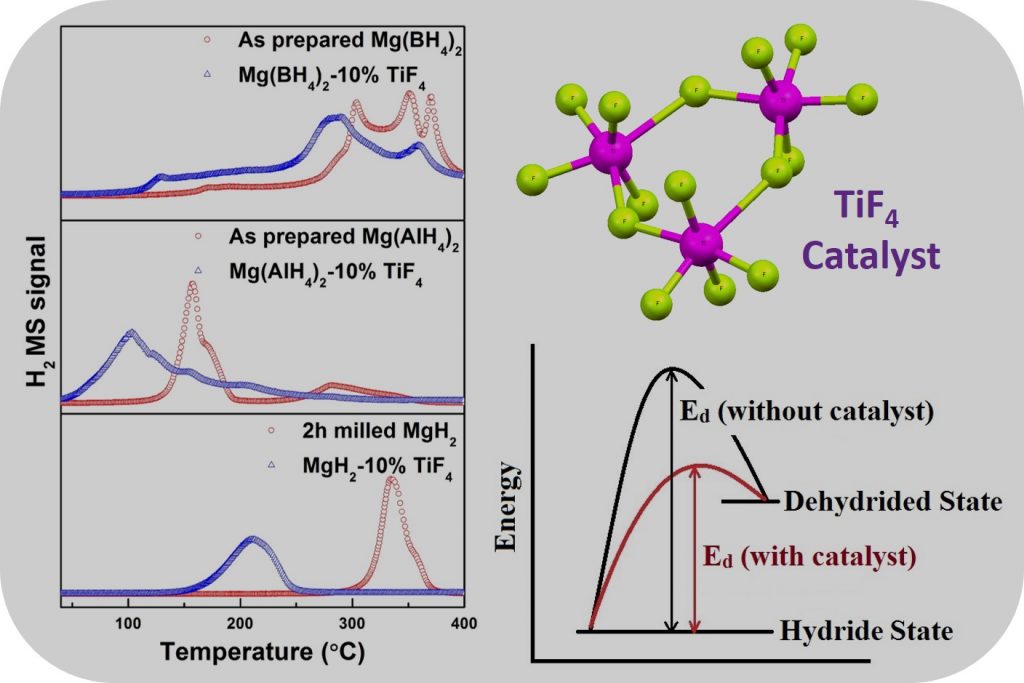
However, the higher working temperatures associated with magnesium and its variants inhibits their practical applicability. There are several efforts devoted to reduce the working temperature and enhance the sorption kinetics using various additives. Titanium-based additives have always been interesting contenders in enhancing the kinetics as well as altering thermodynamics thus reducing the working temperature for various hydrides. Recently, titanium (iv) fluoride has shown superior catalytic activity in the decomposition of the complex variants but still more needs to be done.
Researchers led by Professor Takayuki Ichikawa at Hiroshima University in Japan studied in detail the effects of decomposition of intricate magnesium variants using Titanium (iv) fluoride. The researchers hoped to elucidate the reaction mechanism of the Titanium (iv) fluoride doped magnesium hydride and its complex variants. Their research work is now published in Journal of Materials Chemistry A.
The author observed that the addition of 10% by weight of Titanium (iv) fluoride to the magnesium based hydride materials greatly influenced the decomposition temperature. The decomposition temperature for all three reactions was seen to shift to the lower temperature side. The researchers also noted that the fluorine anion acted as a functional anion when compared to its analogous inactive chlorine anion and in turn significantly improved the decomposition behavior of all the three hydrides.
The Takayuki Ichikawa study has established a mechanism for the decomposition pathway of magnesium hydrides. Using X-Ray diffraction and X-ray photoelectron spectroscopy, the role of Titanium (iv) fluoride has been understood in improving the decomposition properties. The titanium ions have been observed to reduce to lower oxidation states in all the sample used. However, the dead mass comprising of magnesium flouride obtained at the end of the process is still an issue as it slightly lowers the hydrogen content liberated.
Reference
Ankur Jain, Shivani Agarwal, Sanjay Kumar, Shotaro Yamaguchi, Hiroki Miyaoka, Yoshitsugu Kojimaa Takayuki Ichikawa. How does TiF4 affect the decomposition of MgH2 and its complex variants? – An XPS investigation. J. Mater. Chem. A, 2017, 5, 15543–15551
Go To Journal of Materials Chemistry A
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



