Significance
The controlled folding of single polymer chains into well-defined nanoscale architectures represents one of the most compelling frontiers in polymer chemistry. Inspired by the intricate folding of natural proteins, researchers have long sought to design synthetic macromolecules capable of self-organization through intramolecular interactions. Single-chain polymer nanoparticles (SCNPs) have emerged from this pursuit as a powerful class of materials where individual polymer chains collapse into compact entities via covalent or non-covalent linkages. Their structural precision and tunable functionality render them promising candidates for catalysis, sensing, and drug delivery. Yet, achieving rational control over their folding and unfolding processes remains a substantial challenge, particularly when dynamic, reversible interactions are required. Metal coordination offers a versatile means to guide molecular organization, drawing parallels with the role of metal cofactors in biological systems. However, while metal-mediated folding has been explored in small molecules and peptides, its systematic application in synthetic SCNPs is far less developed. Most previous strategies have relied on covalent crosslinking or rigid ligand–metal bonds that limit reversibility. The ability to drive polymer compaction through transient metal–ligand interactions—strong enough to induce collapse, yet labile enough to reverse upon demand—could open an entirely new dimension of structural control in soft materials.
To this account, new research paper published in Polymer Chemistry and conducted by Dr. Sebastian Gillhuber, Dr. Jochen Kammerer, Dr. Ada Quinn, Dr. Joshua Holloway, Kai Mundsinger, Dr. Evelina Liarou, Dr. Dmitri Golberg, Dr. Hendrik Frisch, Dr. Megan O’Mara and led by Professor Christopher Barner-Kowollik from the Queensland University of Technology (QUT) in Australia and Professor Peter W. Roesky from the Karlsruhe Institute of Technology (KIT) in Germany, the researchers developed two complementary models to elucidate Ba(II)-induced polymer compaction. The first is an experimental model showing that coordination between Ba²⁺ ions and polymeric carboxylates produces reversible, intramolecular nanoparticle folding. The second is a molecular-dynamics model capturing the transient, non-permanent nature of these interactions, where barium ions dynamically bridge distant chain segments. Together, they establish a comprehensive picture of reversible, metal-controlled single-chain folding validated by atomic-scale imaging.
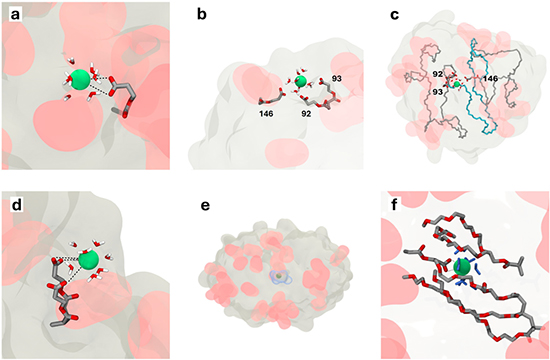
The research team began by synthesizing a statistical copolymer, designated P1, through RAFT polymerization of PEG methyl ether methacrylate and 2-carboxyethyl acrylate. The composition balanced water solubility with a controlled fraction (~14 %) of pendant carboxyl groups, which later served as coordination sites. Upon gradual mixing of an aqueous P1 solution with barium hydroxide octahydrate, intramolecular folding was triggered by complexation between Ba(II) ions and deprotonated carboxylates, producing Ba-functionalized nanoparticles (SCNP1-Ba).
Spectroscopic and hydrodynamic analyses confirmed the transition from an extended coil to a compact entity. Infrared spectra showed the disappearance of the carboxylic C=O stretch and emergence of symmetric and asymmetric COO⁻ vibrations, signaling conversion to barium carboxylates. Diffusion-ordered NMR revealed a measurable increase in diffusion coefficient consistent with reduced hydrodynamic volume, while SEC traces shifted toward lower apparent molar mass, and DLS data indicated a modest but reproducible decrease in particle diameter. Collectively, these results established the successful intramolecular compaction of single polymer chains.
To verify that folding originated from Ba(II) coordination rather than altered hydrogen bonding, a styrene-based analogue lacking polar groups was tested; it too underwent barium-induced compaction, confirming the metal-driven mechanism. Complementary MD simulations provided a dynamic picture of these interactions. Over simulated trajectories, barium ions continuously associated and dissociated from polymer carboxylates, spending roughly two-thirds of their time coordinated yet rarely forming permanent inner-sphere complexes. Instead, the dominant motif involved transient bridging within the second coordination shell, wherein one ion simultaneously contacted multiple carboxylates. This produced momentary intramolecular crosslinks that collectively reduced polymer dimensions without rigidifying the structure. The simulations further revealed that Ba(II) accelerated the collapse of extended chains but did not substantially change the equilibrium size, underscoring a kinetic—rather than purely thermodynamic—role in compaction. The authors demonstrated the reversibility of folding by adding sulfuric acid to precipitate insoluble BaSO₄, thereby removing the coordinating ions and regenerating the original carboxylic acid polymer. SEC and IR spectra of the recovered material were nearly identical to those of the starting P1, and a subsequent re-addition of Ba(II) faithfully re-established the SCNP state. Finally, the heavy-atom contrast of barium enabled direct imaging of individual metal atoms by annular-dark-field STEM. At low magnification, bright clusters marked Ba-rich regions, while at atomic resolution, discrete barium atoms appeared as distinct luminous points. Quantitative image analysis revealed an average of roughly 30 ions per nanoparticle, though cryogenic blotting techniques suggested that isolated SCNPs often contained only one or two Ba atoms, confirming the high sensitivity of preparation conditions. This atomic-level visualization of metal distribution within single polymer chains marks a technical milestone for metallopolymer characterization.
In conclusion, the collaborative research work of Professor Christopher Barner-Kowollik and Professor Peter W. Roesky with their groups, redefines how dynamic metal–polymer interactions can be harnessed to engineer adaptive nanostructures. By exploiting the transient binding of Ba(II) ions, the researchers demonstrated a system where single-chain nanoparticles can fold and unfold reversibly under mild, aqueous conditions. Such control transcends conventional covalent or supramolecular strategies, offering a non-destructive handle over polymer conformation analogous to biological metalloproteins that modulate structure through metal-ion exchange.
The implications extend beyond the specific case of barium. Because the coordinating 2-carboxyethyl groups mimic the side chain chemistry of glutamic acid, the polymer backbone serves as a synthetic analogue for natural systems where Ca²⁺ or Mg²⁺ ions govern folding or catalysis. Thus, the SCNP1-Ba model provides a minimal yet experimentally tractable framework for investigating the thermodynamics and kinetics of metal–carboxylate interactions in biomimetic environments. Moreover, the observed reversibility through BaSO₄ precipitation underscores a general strategy for controlling polymer states by selective removal or introduction of counter-ions—a concept that may inspire switchable materials, recyclable catalysts, or stimuli-responsive delivery carriers.
Equally important is the methodological advance achieved through atomic-level STEM imaging. The ability to detect individual heavy-metal atoms within single polymer chains bridges a long-standing analytical gap, allowing researchers to quantify metal distribution, correlate structure with catalytic or optical properties, and validate computational models with direct visual evidence. When coupled with MD simulations, this dual experimental–computational approach establishes a blueprint for probing dynamic metallopolymer systems that cannot be described by static spectroscopy alone. In a broader context, the study demonstrates that intramolecular compaction does not necessitate permanent crosslinking but can emerge from a dynamic equilibrium of transient coordination events. This insight may influence future design principles in soft-matter chemistry, where achieving mechanical integrity and reversibility often stand at odds. The authors’ integration of reversible metal binding, precise polymer synthesis, and high-resolution imaging thus offers a platform for exploring catalysis, sensing, and self-repair mechanisms at the single-chain level. Ultimately, the work exemplifies how collaboration across synthetic, computational, and analytical disciplines can resolve long-standing questions about structure–function relationships in metallopolymers. The results invite extensions to other ions, ligands, and solvent environments, opening pathways toward artificial materials that emulate the subtle responsiveness of biological macromolecules.
Reference
Gillhuber, Sebastian & Kammerer, Jochen & Quinn, Ada & Holloway, Joshua & Mundsinger, Kai & Liarou, Evelina & Golberg, Dmitri & Frisch, Hendrik & O’Mara, Megan & Barner-Kowollik, Christopher & Roesky, Peter. (2025). Control over Ba( ii )-mediated single-chain polymer nanoparticle compaction by dynamic metal complexation. Polymer Chemistry. 16. 10.1039/D5PY00883B.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



