Significance
The demand for higher capacity and longer lifetimes in lithium-ion batteries keeps rising, largely pushed by electric vehicles and the sheer number of portable devices we now depend on, researchers have repeatedly circled back to silicon as a potential game-changer for the anode and considered almost ideal: abundant, inexpensive, and able to store a remarkable amount of lithium. However, during lithiation and delithiation it expands far more than the surrounding electrode can comfortably accommodate, and this expansion breaks things in ways that are difficult to reverse. Conductive pathways crack, particles drift apart, and the interphase becomes unpredictable. Before long, the cell’s performance unravels. Traditional slurry-cast electrodes do not help the situation. They introduced binders, conductive agents, and the metal current collector, each one adding mass while contributing little to the battery’s actual function. Worse, these components deform differently than silicon, so the whole assembly becomes mechanically disjointed once cycling begins. This has pushed many groups toward self-supporting Si/C structures that avoid binders altogether and behave more like coherent materials rather than fragile mixtures. Several approaches have been tried, from graphene films to tangled nanotube scaffolds. These designs certainly improve conductivity, but most fall short when silicon starts to move. Either the pores collapse, or the silicon escapes confinement, and the system fractures. The real challenge is to build a structure that can breathe with the silicon without falling apart—something porous yet firm, flexible yet not fragile. That balance has proven surprisingly difficult to achieve.
To this end, new research paper published in Electrochimica Acta and conducted by Professor Quan Li’s team from Institute of Advanced Materials and School of Chemistry and Chemical Engineering at Southeast University. They created a self-supporting Si/C anode built from a bubble-derived 3D graphene-like carbon scaffold capable of confining silicon’s volume expansion while maintaining strong electrical connectivity. They introduced two functional layers: ultrathin nanoscale Si films deposited uniformly through Low-Pressure Chemical Vapor Deposition (LPCVD), and a conformal amorphous carbon overlayer that stabilizes the interface and enhances conductivity. The combination of controlled compression, continuous carbon lamellae, and confined Si films yields exceptional cycling stability and high-rate capability. This architecture demonstrates a practical, scalable pathway for integrating high-capacity silicon into long-life lithium-ion batteries.
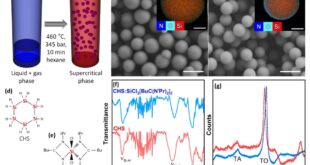
The research team started with formation of the porous carbon framework. A molten organic precursor is rapidly heated, causing volatile gases to expand and produce a foam-like carbon network composed of thin, interconnected sheets. After carbonization, this network retains its bubble-derived morphology, offering a combination of rigidity and accessible voids. The authors adjust the spacing between the carbon lamellae through controlled mechanical compression, enabling a systematic examination of how electrode density and pore accessibility affect performance. Onto this carbon scaffold, they introduced silicon through low-pressure chemical vapor deposition. Rather than forming isolated particles, the silicon condenses into uniform nanoscale films that coat the internal surfaces of the carbon network. This configuration ensures intimate electrical contact throughout the electrode while avoiding the weak interfacial bonding that often plagues particulate composites. A subsequent carbon deposition step produces a thin amorphous layer that envelops the silicon, reinforcing the mechanical interface and protecting it from oxidation. Throughout these steps, the three-dimensional character of the scaffold remains intact, and microscopy shows that the films adhere closely without collapsing the underlying structure.
Afterward, the authors conducted electrochemical evaluation using cyclic voltammetry, which reveals that the coated silicon layer quickly establishes a stable lithiation–delithiation profile after the initial cycle. This early stabilization reflects the combined influence of the conductive carbon backbone and the protective amorphous carbon shell, both of which help regulate interfacial reactions. Moreover, rate testing demonstrates that the composite maintains substantial capacity even at elevated current densities, with the most balanced compression condition providing the most consistent performance. They found electrodes produced with insufficient compression allow greater structural breathing but suffer from poor confinement of silicon. In contrast, heavily compressed samples restrict pore access and hinder ion transport, resulting in premature fading. The intermediately compressed electrode achieves a balance between these competing factors, preserving structural integrity while maintaining electrolyte accessibility. Long-term cycling highlights this difference decisively. The optimally compressed electrode undergoes only modest thickness changes over extended cycling, indicating that the carbon framework effectively restrains silicon expansion. Less optimal samples either swell excessively or exhibit mechanical fractures, confirming that both under‐ and over‐compression compromise durability. The team also performed impedance and diffusion assessments, and found that the moderated structure provides the most favorable ion transport pathways and the least interfacial resistance. Additionally, testing in full-cell configuration further demonstrates that the composite maintains stable operation when paired with a commercial cathode, underscoring its practical viability.
We believe the new composite reflects a shift away from nanostructuring as an end in itself and toward controlling mesoscale architecture—spacing, lamellar coherence, and stress distribution. The 3 MPa compression point becomes a reminder that electrochemical performance is often governed by a delicate balance between confinement and permeability. Too little compression leaves Si inadequately supported; too much suffocates ion flow and stiffens the system beyond what volume changes can tolerate. The researchers situate their design precisely in this middle ground, where mechanical compliance, ion accessibility, and electronic connectivity reinforce each other. This balance is rarely achieved so cleanly in Si-based anodes, where improvements in one domain often degrade another.
In conclusion, the findings of Southeast University scientists highlight the practical benefits of creating an electrode that is intrinsically self-supporting. Eliminating binders and conductive additives removes several variables that typically complicate electrode degradation. The resulting composite behaves as a unified material rather than a collection of loosely integrated components. The amorphous carbon overlayer plays a quiet but essential role, buffering surface defects, stabilizing SEI formation, and enhancing conductivity without disrupting structural coherence. These design choices collectively reduce irreversible lithium losses, yielding a notably high initial coulombic efficiency for a silicon-rich framework.
Another important advantage is scalability and the bubble-derived porous graphene-like carbon (PGN) scaffold is formed through a straightforward thermal expansion process that is inherently rapid and does not depend on highly specialized templates. Compression, carbonization, and LPCVD—while requiring careful optimization—are all processes compatible with existing battery manufacturing infrastructure. This practicality gives the work a broader relevance beyond academic demonstration. If such self-supporting composites can be produced in larger formats with consistent thickness and uniform Si loading, they could address persistent barriers to commercializing high-capacity silicon anodes. The limited thickness expansion observed over prolonged cycling suggests that these electrodes could be integrated into full-cell stacks without necessitating excessive safety margins in cell casing or electrolyte dosing. The results also hint at intriguing possibilities for next-generation architectures. The laminated carbon–silicon–carbon configuration resembles a controlled confinement environment where mechanical, electronic, and chemical interactions can be tuned independently. One might imagine varying carbon film thickness, substituting alternative amorphous carbon precursors, or incorporating dopants to further improve conductivity. The 3D nature of the framework implies that more elaborate gradient or multi-layer designs could be achievable without compromising mechanical stability. In a nutshell, the authors’ new approach offers a compelling direction for future battery materials research, where structural intelligence becomes as central as chemical composition.
Reference
Junwei Shan, Zhiqing Zhu, Zhihao Chen, Feng Du, Quan Li, Si/C composites based on bubble 3D graphene-like porous materials as self-supporting anodes for high-performance lithium-ion batteries, Electrochimica Acta, Volume 529, 2025, 146281,
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.