Significance
Covalent organic frameworks (COFs) have attracted a lot interest because they promise something that is hard to achieve in porous materials which is crystallinity without sacrificing molecular design freedom. In principle, assembling extended frameworks from light, organic building blocks allows chemists to control pore dimensions, connectivity, and surface chemistry almost at will. That level of tunability explains why COFs continue to appear in discussions of gas separations, heterogeneous catalysis, optoelectronics, and, more recently, resource recovery. In practice, however, making these materials is not straightforward and a large fraction of reported COFs still rely on slow, reversible condensation reactions that require days of heating, carefully chosen solvent systems, or narrowly optimized conditions. These approaches are workable, but they are rarely efficient. Atom economy is frequently poor, byproducts are unavoidable, and scaling beyond small laboratory batches remains challenging, which has limited broader adoption.
β-Ketoenamine-linked COFs are often cited as an exception when chemical stability is the primary concern. Their resistance to acidic and basic environments is well documented and is generally attributed to tautomeric stabilization combined with strong intramolecular hydrogen bonding. Compared to imine-linked frameworks, they tend to survive conditions that would otherwise lead to rapid degradation. Ironically, this same stability makes them harder to synthesize well. Conventional routes typically involve multistep condensations carried out over long reaction times, where crystallinity is achieved only at the expense of synthetic efficiency. As a result, the field has largely accepted an implicit compromise: robust frameworks are possible, but only if one is willing to tolerate slow, resource-intensive synthesis.
Against this backdrop, click chemistry offers a fundamentally different way of thinking about framework construction. Click reactions are designed to be fast, selective, and atom-economic, and their extension to polymerization has already reshaped several areas of materials chemistry. Amino-yne click polymerization, in particular, stands out because it proceeds without metal catalysts, theoretically 100% atom-economic, and forms dynamically reversible bonds that can correct structural errors during growth. Despite these advantages, it has rarely been explored as a viable strategy for COF synthesis, especially for chemically resilient β-ketoenamine frameworks. This approach has been relatively underexplored, and it raises a simple but important question: whether synthetic efficiency and framework robustness truly need to be mutually exclusive.
To this end, new research paper published by Professor Anjun Qin et al. report a metal-free amino-yne click polymerization strategy for constructing β-ketoenamine-linked covalent organic frameworks. This approach enables highly crystalline COFs to form within hours while preserving structural robustness. The resulting materials combine exceptional gold-ion adsorption and in situ reduction with ultralong organic room-temperature phosphorescence.
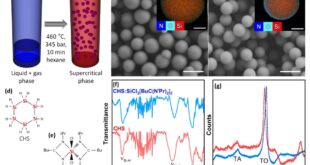
The research team constructed two-dimensional COFs through the reaction of a trifunctional, carbonyl-activated ethynyl monomer with structurally distinct triamine nodes. Under solvothermal conditions, amino-yne click polymerization proceeded rapidly, completing framework formation within hours rather than days. The reaction environment was deliberately simple, avoiding metal catalysts and minimizing auxiliary reagents, yet it proved sufficient to drive long-range ordering. Structural characterization confirmed the formation of well-defined crystalline frameworks with uniform pore architectures and complete conversion to β-ketoenamine linkages. Both materials exhibited permanent microporosity and high surface areas. These structural features translated into functional performance. The COFs showed high adsorption capacities for Au(III) ions from aqueous solution, with rapid uptake kinetics and pronounced selectivity over competing transition metal ions. Post-adsorption analyses further revealed partial in situ reduction of Au(III) species within the frameworks, which may contribute to sustained performance over multiple cycles. In addition, exfoliation of the bulk COFs into nanosheets and their incorporation into a rigid polymer matrix activated ultralong room-temperature phosphorescence, with afterglow lifetimes extending into the hundreds of milliseconds. This behavior, which remains challenging for purely organic systems, is attributed to the combined effects of carbonyl-induced spin-orbit coupling and hydrogen-bond-reinforced structural rigidity within the β-ketoenamine network.
In conclusion, the new work established a versatile and sustainable platform for multifunctional COF design and demonstrated successful that amino-yne click polymerization can serve as a direct route to crystalline β-ketoenamine-linked COFs. Indeed, the ability to obtain highly ordered COFs within hours, using metal-free and atom-economic chemistry, represents a meaningful step toward sustainable materials synthesis. The results further demonstrate that dynamic covalent chemistry can accommodate rapid framework formation while maintaining structural order. The reversible nature of amino-yne bond formation enables error correction during framework growth, preserving crystallinity even under accelerated conditions and the new findings may inspire the exploration of other underutilized click reactions as COF linkages, broadening the design space beyond traditional imine, boronate, or hydrazone chemistries. Additionally, we think the exceptional performance in gold capture positions these materials at the forefront of COF-based metal recovery and the adsorption capacities the authors reported rival or exceed many existing frameworks, especially those lacking sulfur-containing motifs. More importantly, the demonstrated selectivity under competitive conditions speaks directly to real-world relevance, where precious metals are typically present in complex mixtures. The intrinsic ability of the frameworks to reduce Au(III) to metallic gold adds another layer of value, effectively coupling separation with transformation and simplifying downstream processing. The photophysical findings extend the implications even further. Ultralong organic room-temperature phosphorescence remains a challenging goal due to the inherent instability of triplet states in purely organic systems. Furthermore, the authors provided a new structural paradigm for stabilizing long-lived excited states by leveraging the rigidity, hydrogen bonding, and electronic characteristics of β-ketoenamine COFs,. This approach highlights the potential of COFs to serve not only as structural scaffolds but also as luminescent materials with tunable emission properties. Together, these findings provide design insights for developing COFs that integrate structural order with functional performance relevant to metal separation and luminescent applications.
Reference
Bai, Wei & Song, Bo & Tian, Qi & Fu, Xinyao & Qin, Anjun & Tang, Ben. (2025). Efficient and Atom-Economic Construction of Functional β-Ketoenamine-Linked Covalent Organic Frameworks via Amino-yne Click Polymerization. Chemistry of Materials. 37. 10.1021/acs.chemmater.5c00549.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.