Significance
Product quality is paramount in pharmaceutical industries for guaranteeing the reproducible delivery of the therapeutic benefits explicated in the label. Today, both in pharmaceutical and biological industries, assurance of product quality and safety is based on the Quality-by-Design paradigm. This initiative states that final product quality should be built-in during the process and not merely tested as previously done. To this end, the product and process design should be based on the understanding of the physical phenomena taking place during the process and of the interaction between process and product.
In pharmaceutical industries, freeze drying is widely used as preservation method for heat sensitive products, such as vaccines. This drying process comprises three main steps: freezing, devoted to the formation of ice crystals and the solidification of the product; primary drying, during which ice crystals are removed by sublimation; secondary drying, during which the adsorbed water is removed from the product until the target value of residual moisture content is achieved.
The position of the vial on the shelf of the equipment may affect the effectiveness of the freeze-drying process, as it directly impacts the product temperature value. Temperatures above a critical value compromise the porous structure of the product, resulting in its collapse and rejection. It is possible to classify two groups of vials in function of their position on the shelf: the “edge vials” are those located on the shelf periphery, whereas “central vials” are located in the center of the shelf and not directly exposed to the chamber door and walls. Vials placed at the edges of the shelves may experience higher temperatures as compared to those placed at the center and could have a higher rejection risk.
The “edge vial effect”, as it is typically referred to, represents a significant issue in vial freeze-drying. Most of the experimental studies conducted so far have difficulties in determining the exact mechanisms causing the edge vial effect. Consequently, proposing effective heat transfer predictions for freeze-drying cycle design and scale-up has been a challenge.
A team of researchers from INRA and AgroParisTech at Université Paris-Saclay in France and GlaxoSmithKline Biologicals SA in Belgium tackled this challenge: Bernadette Scutellà and colleagues developed a detailed 3D mathematical model for analyzing heat transfer in the vials. Their main aims were the prediction of the heat flows in vials differently located on the shelf and the understanding of the role played by the different heat transfer mechanisms, thus to provide an efficient prediction of the edge vial effect. This work was published in the journal Applied Thermal Engineering.
The authors used COMSOL Multiphysics to solve numerically the 3D model of the heat transfer during the second step of the freeze-drying process, i.e. primary drying. The simulation represented a part of the freeze-drying chamber with five vials strategically located at the edges of the shelves in contact and not in contact with a metallic rail. Heat transfer mechanisms considered in the numerical experiments included: radiation, evaluated by the hemi-cube method taking into consideration the shadowing effect in the system; gas conduction near solids, evaluated by simulating a fictitious layer built at the interface of the solid and the gas; contact conduction between the solids, simulated as proposed in previous study of Scutellà et al. (2017a).
The numerical predictions were validated against experimental measurements of heat flow rates received by edge and central vials at different chamber pressure and shelf temperature values, which corroborated the overall predictive capacity and accuracy of the mathematical model especially for the cases relevant to pharmaceutical applications. By using the developed mathematical model, the authors successfully predicted the heat flows received by the five vials located at the edge and in the center of the shelves.
Among the three major heat transfer mechanisms investigated in the study, gas conduction proved to be the leading cause of higher heat transfer at the edges than in the center. Furthermore, the rail contributed more to the radiation heat transfer than the walls. These findings were original, as previous literature attributed the edge vial effect mainly to the radiation from the drying chamber door and walls.
The proposed model is versatile and can be used in predicting the edge vial effect for various loading configurations and equipment characteristics. The information obtained in such cases will help in designing efficient freeze-dryer process which will, therefore, enhance the quality of the product.
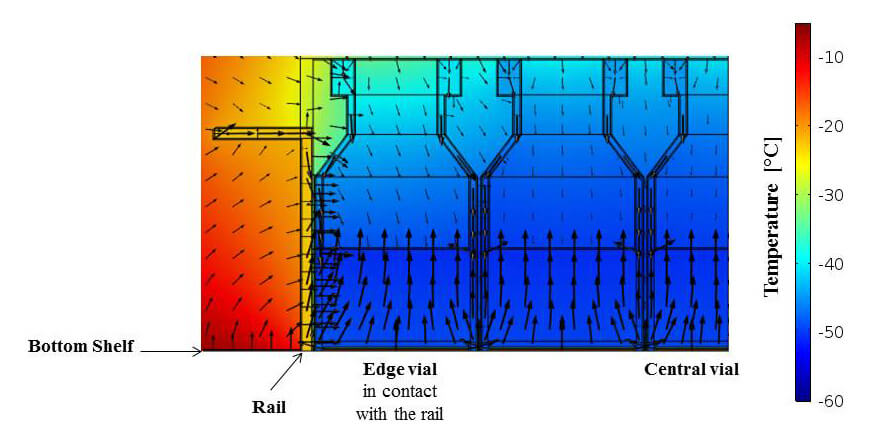
Temperature profiles and heat fluxes in the edge vial in contact with the rail and in the central vial at a shelf temperature of 0 °C and a chamber pressure of 4 Pa. Arrow length indicates flux magnitude on a logarithmic scale, to improve the visualization of smaller fluxes from top, wall and rail.
Figure adapted from Scutellà, B., Plana-Fattori, A., Passot, S., Bourlès, E., Fonseca, F., Flick, D., & Tréléa, I. (2017b). 3D mathematical modelling to understand atypical heat transfer observed in vial freeze-drying. Applied Thermal Engineering, 126, 226-236, with permission from Elsevier.
References
Scutellà, B., Passot, S., Bourlès, E., Fonseca, & Tréléa, I. (2017a). How vial geometry variability influences heat transfer and product temperature during freeze-drying. Journal of Pharmaceutical Sciences, 106:3, 770-778..
Go To Journal of Pharmaceutical Sciences
Scutellà, B., Plana-Fattori, A., Passot, S., Bourlès, E., Fonseca, F., Flick, D., & Tréléa, I. (2017b). 3D mathematical modelling to understand atypical heat transfer observed in vial freeze-drying. Applied Thermal Engineering, 126, 226-236. .
Go To Applied Thermal Engineering Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.