Significance
Methane is a critical greenhouse gas, significantly contributing to atmospheric warming. The global rise in methane levels, from 729 ppb in 1750 to 1910 ppb by 2022, has resulted in a marked increase in anthropogenic radiative forcing. Agricultural activities, particularly animal production, account for a substantial portion of methane emissions. Conventional methane sinks, such as soil methanotrophs and gas-phase radical reactions, are inadequate in balancing this increase. Historically, methane oxidation technologies, such as regenerative thermal oxidation and catalytic thermal oxidation, have been effective for high-concentration methane streams. However, these methods are impractical for low-concentration sources due to high costs, energy demands, and byproduct formation, such as NOx gases. Additionally, biofilters, photocatalysts, and catalysts combined with zeolites, though emerging, have yet to be proven effective at scale.
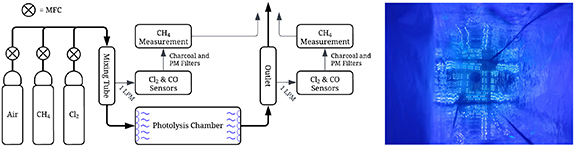
A new study published in Environmental Research Letters by Morten Krogsbøll, Dr. Hugo Russell and Dr. Matthew Johnson from University of Copenhagen and Ambient Carbon ApS in Denmark, embarked on an innovative project to address the issue of methane removal, particularly at low concentrations which is a significant challenge in environmental management. The authors introduced a novel methane eradication photochemical system (MEPS) that utilizes chlorine atoms in the gas phase to oxidize methane efficiently. The researchers developed a laboratory prototype of a new system for methane removal, using chlorine atoms to initiate gas-phase reactions. They tested this prototype under controlled conditions, focusing on its ability to remove methane from waste air. They examined key parameters like the chlorine to methane ratio (Cl2:CH4), UV light power, and reactor design to optimize the removal efficiency.
The designed laboratory prototype achieved a 58% removal efficiency for methane at a concentration of 55 ppm, which is significant considering the low concentration levels. The system showed a promising range of energy consumption (36 to 244 kJ m−3), indicating its efficiency in terms of energy use compared to existing methane removal methods. The apparent quantum yield (AQY) ranged from 0.48% to 0.83%, providing insight into the efficiency of photon utilization in the process. The findings suggest that MEPS has the potential for scalability and could be applied in various settings, including agricultural and industrial scenarios, where low-concentration methane emissions are prevalent. The system operates under ambient conditions, which reduces energy requirements and potential environmental impacts compared to traditional methane oxidation methods.
The MEPS technology introduces a novel approach, leveraging chlorine atoms in gas-phase reactions to oxidize methane. The system operates under ambient conditions, which significantly reduces the energy requirements compared to traditional methods. This system’s effectiveness in oxidizing methane at concentrations as low as 50 ppm marks a significant advancement. Energy consumption is a critical factor in evaluating methane oxidation technologies. MEPS demonstrates an energy-efficient approach, with a specific energy use of 2.1–7.7 kWh/g of methane. These figures are promising, particularly when compared to the energy requirements of traditional methods. The adaptability and scalability of MEPS are significant. Its potential applications in agricultural settings and wastewater treatment plants could address a substantial portion of methane emissions. The ongoing development and planned field tests are crucial steps towards implementing this technology on a larger scale. The environmental impact of MEPS extends beyond methane removal. The system’s low energy requirement and potential integration with renewable energy sources could contribute to a broader sustainable strategy. Economically, the feasibility of MEPS in reducing methane emissions at a lower cost than existing technologies could be a game-changer, particularly in sectors where methane emissions are a significant concern.
Further development of MEPS is essential. Optimizing the reactor design, enhancing the chloride recycling system, and integrating advanced light sources are areas for future research. The upcoming field tests will be critical in determining the system’s effectiveness in real-world conditions and its potential scalability. In summary, their research marks a significant advancement in the field of environmental engineering, offering a new, efficient, and potentially scalable method for reducing low-concentration methane emissions, a key contributor to global warming.
Reference
Morten Krogsbøll, Hugo S Russell and Matthew S Johnson. A high efficiency gas phase photoreactor for eradication of methane from low-concentration sources. Environmental Research Letters, 2023, Volume 19, Number 1
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



