Significance Statement
Convectional thermodynamic theories fail to achieve capillary condensation since it involves an activated process. This promotes the application of transition state theory in which the rate constant of capillary condensation in open ended mesoporous materials is estimated as the probability of overcoming the energy barrier. The kinetic nature of capillary condensation described by the theory should offer a new interpretation of the phase transition in confined space, which leads to the comprehensive characterization of the mesoporous materials with regard to pore size and porosity.
In a recent work published in ACS Nano, scientists from the Department of Chemical Engineering at Kyoto University in Japan studied the mechanism to kinetically control capillary condensation in nanopores.
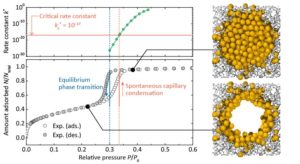
Capillary condensation happens at pressures higher than capillary evaporation pressure in open ended mesopores which lead to an adsorption hysteresis. The researchers started with argon adsorption recreations in cylindrical even pore models of several pore dimensions and atomistic silica pore models that mimic known mesopores using the grand canonical Monte Carlo and gauge cell Monte Carlo methods. Subsequently, dimensionless frequency constants for the capillary condensation in the silica pore models were tested using the transition state theory.
It was observed that in all the examined pore models, the capillary condensation in the grand canonical Monte Carlo simulation occurs at the chemical potential where the dimensionless frequency constant is equal to the critical dimensionless rate constant without having any interdependence with the capillary condensation regime. They also noted that the critical dimensionless frequency constant was equal independently of the variety of the pore models employed in the simulation. These availed ways to estimate capillary condensation for the grand canonical Monte Carlo system at all examined temperatures over a diverse range of varying mesopore sizes and determine the boundary separating the reversible condensation (no adsorption hysteresis) regime and the developing hysteresis regime.
In the grand canonical Monte Carlo simulation, smooth cylindrical pores of infinite length along the pore were assumed. For these type of pores, formation of cavity in the pore, which is the activated process, was observed. From the grand canonical Monte Carlo simulation, it was realized that the maximum density fluctuations were smaller than for realistic systems. The team concluded that the critical rate constants controlling the capillary condensation and evaporation in the grand canonical Monte Carlo simulation were similar. Also, the vapor-liquid phase transition in the thermodynamic equilibrium did not occur when the rate constant is smaller than the critical rate constant in confined space.
Thorough comparison between the experimental and simulated adsorption of isotherms on the atomistic silica pore models indicate that the frequency constant of the real system also has a universal value, thereby revealing that the critical rate constant can be applied over an extensive range of temperatures and pore sizes to quantitatively estimate the experimental capillary condensation pressures.
The calculated pressure dependences of the rate constant also revealed that the experimental equilibration time hardly affects the resulting capillary condensation pressures. This agrees with the empirical fact that identical adsorption isotherms are always observed even though the experimental conditions slightly differ depending on research institutes and experimental apparatuses. The researcher concluded that, for example, about a million years are needed as the equilibration time to observe the reversible (no adsorption hysteresis) isotherm for MCM-41 with pore diameter of 3.9 nm at 75 K.
Reference
Tatsumasa Hiratsuka, Hideki Tanaka, and Minoru T. Miyahara. Mechanism of Kinetically Controlled Capillary Condensation in Nanopores: A Combined Experimental and Monte Carlo Approach. ACS Nano, 2017, 11 (1), pp 269–276.
Department of Chemical Engineering, Kyoto University, Katsura, Nishikyo, Kyoto 606-8501, Japan.
Go To ACS Nano Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



