Significance
Body organs and often subjected to large external mechanical forces that, if not properly controlled, could damage the organs or paralyze normal body operations. During walking, for example, the Achilles tendon experiences a mechanical force greater than the body weight. Through homeostasis, a self-regulating process for biological systems, body organs, and systems can sufficiently regulate the impact of external mechanical forces. Weak mechanical stimulation is also crucial in driving tissue homeostasis as it prevents the loss of muscle masses. The significance of mechanical stimulation in tissue functions is further linked to the effects of mechanical stimuli, such as matrix topology and matrix deformation, on the behavior of cells and tissues. Several studies to establish the effects of mechanical stimuli on cell differentiation and proliferation have been carried out. The results revealed that mechanosensing is mostly regulated by extracellular matrix (ECM) proteins like fibronectin and collagen, which bind with adherent cells. Nevertheless, subsequent studies have established that mechanosensing cannot be sufficiently regulated by ECM alone due to the inherent complexity of cellular response to mechanical stimuli.
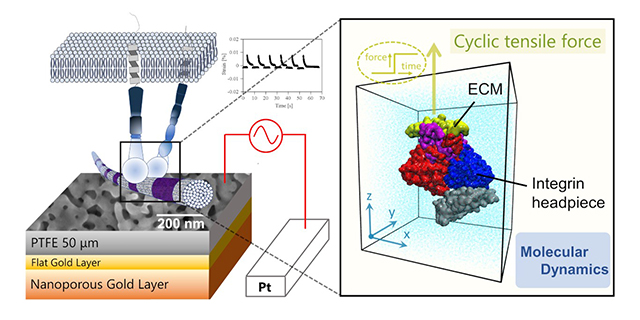
Generally, cells are anchored to the extracellular matrix through proteins, integrins in particular. This shows the indispensable role of integrins in mechanotransduction/mechanosensing, which is mainly based on the type and abundance of the integrins. Being a key regulating factor, the impact of different types of integrin has been thoroughly studied. Recently, molecular dynamics simulations revealed that active integrins have a hinge-opening structure between the βA and hybrid domains, which binds with ECM to cause swingout motion. The integrin changes due to binding with ECM are broadly classified into two: αI integrin types and αI-less integrin type, which have been hypothesized to influence mechanosensing significantly. This hypothesis can only be accurately tested if the transfer of mechanical stimulus via molecules other than integrins is effectively suppressed. Unfortunately, it is challenging to achieve using available scaffolds such as silicone and hydrogels.
Nanoporous metals, specifically nanoporous gold actuators, are promising alternatives to scaffolds because they produce desirable charge-induced formation due to large specific surface areas. Moreover, effective tissues cells strongly adhere to gold surfaces due to bind gold and amino acids effectively. Equipped with this knowledge, Kyoto University researchers: Soichiro Deguchi (PhD candidate), Mr. Atsushi Kato, Mr. Peizheng Wu, Professor Masataka Hakamada, and Professor Mamoru Mabuchi tested the above hypothesis through molecular dynamic simulation experiments to study the effects of cyclic mechanical stimulus on cell proliferation. Specifically, nanoporous gold (NPG) actuator was utilized to prevent mechanical stimulus transfer via molecules other than integrins without using a scaffold. Their research work is currently published in the research journal, Acta Biomaterialia.
Results showed that only a cyclic strain of 0.5% was generated by the NPG actuator to load the mechanical stimulus without scaffolds. This cyclic strain, though relatively small, was sufficient to improve the proliferation of fibroblasts. This indicated the capability of cells to sense such small strains with the help of integrins. α5β1 and αvβ3 integrins exhibited high mechanosensitivity than α1β1 and α2β1 integrins. Similar results were confirmed with experiments involving integrin inhibitors. The differences in the conformal changes of the integrin headpiece due to binding with ECM resulted in the difference in mechanosensitivity between the two integrin types.
In summary, the authors proved the hypothesis linking mechanosensing with integrin type by using NPG to restrict the transfer of mechanical stimulus to only integrin molecules without scaffolds. The simulation results suggested that αI integrins and αI-less integrins have low and high mechanical sensitivity, respectively. In doing so, the study also confirmed the heterogeneous roles of integrins in facilitating mechanosensing. In a statement to Advances in Engineering, Professor Mamoru Mabuchi said the study will advance future research on mechanical stimulation of cell behaviors for homeostasis.
Reference
Deguchi, S., Kato, A., Wu, P., Hakamada, M., & Mabuchi, M. (2021). Heterogeneous role of integrins in fibroblast response to small cyclic mechanical stimulus generated by a nanoporous gold actuator. Acta Biomaterialia, 121, 418-430.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




