Significance
Since the emergence of nanotechnology, its advancement has positively impacted different fields. For instance, advances in nanotube synthesis and functionalization have paved the way for intensive research on mass transport through materials with nanochannels thus enabling experimental realization of initially formulated theories. Unfortunately, despite the establishment of hydrodynamics, the fluid transport phenomena have not been fully explored. Thus, with the rapid increase in the applications of nanofluidic technology, effective methods for the fabrication of nanoscale fluidic systems are highly desirable. This requires the understanding of the behavior of the interfacial water and particularly the interaction between the water molecules and nanotube walls that plays a major role in the fluid-structure and dynamics.
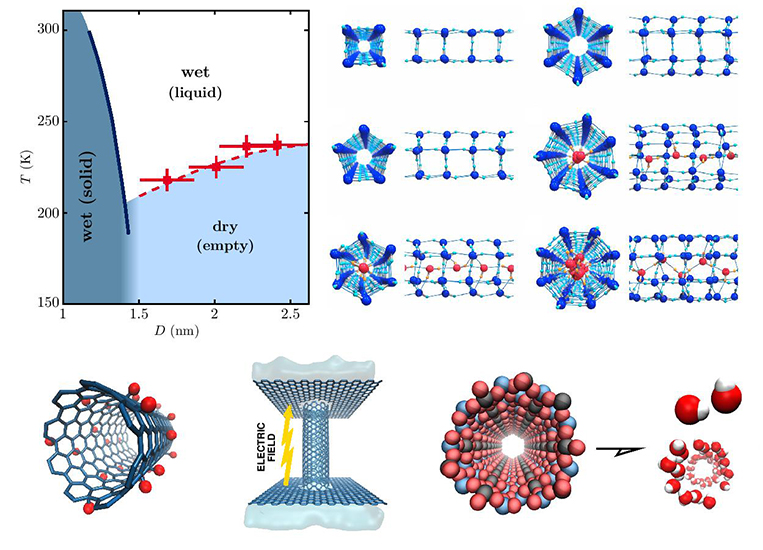
Regarding this, scientists from the Universidade Federal do the Rio Grande do Sul: Dr. Mateus Köhler, Dr. José Bordin, Dr. Carolina de Matos and Professor Marcia Barbosa looked carefully on the impact of hydrophobicity over the nanopore water transport that has exhibited potential influence on the fluid organization and mobility. Through the help of nanotubes functionalized with hydrophobic and hydrophilic sites, they also studied the effect of polarity heterogeneity over confined water properties. Their main objective was to explore the changes in the interaction and confining distance between the water molecules and nanotube walls and their effects on freezing, wet/dry transitions and flow rates properties. Their work is currently published in the research journal, Chemical Engineering Science.
Briefly, the research team analyzed two aspects that impact on the behavior of confined water. This included the structural characteristics of various confinements and their influence on the fluid mobility as well as the physical-chemical mechanisms exhibited by the water confined in both polar and nonpolar nanotubes.
For water confined in nanotubes, the authors noted that their flow and boundary conditions are determined by the intermolecular attractions between water and the nanopore walls. Thus, a change in the water dynamics concerning the nature of the nanotube wall i.e. hydrophobic, hydrophilic or a mixture of the two was proposed. They also noted that polarity was a key parameter in structuring of water inside nanopores that further determined the mobility of confined water. This also helped in the tuning of the water-nanotube interaction through increasing the hydrophobic or hydrophilic nature thus obtaining desirable structural and dynamical transitions.
The study identified nanofluidic through functionalized polar and nonpolar nanotubes as a flourishing field with vast opportunities. Therefore, the ability to control the water flow in nanotubes as described in the paper is of great significance in fabricating highly efficient nanofluidic devices with numerous applications such as contaminant selectivity, energy conversion, and desalination processing.
Even though the study identified some challenges to overcome, Dr. Köhler the corresponding author in a statement to Advances in Engineering explained the importance of the study in enhancing large scale application of nanofluidic. This is because it allows for the design of well-controlled functionalized nanotubes with improved accuracy in measurement of the water flow inside the pores.
Reference
Köhler, M., Bordin, J., de Matos, C., & Barbosa, M. (2019). Water in nanotubes: The surface effect. Chemical Engineering Science, 203, 54-67.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


