Significance
All-solid-state lithium metal batteries are central for future energy storage, because they have very high energy density and high safety compared to liquid electrolytes. Within this broader effort, polymer-based solid electrolytes, especially those built around poly(ethylene oxide) (PEO), have never really left the conversation. PEO systems are attractive in a very practical sense. They are mechanically soft, easy to process at scale, and chemically compatible with many lithium salts. These are not trivial advantages, particularly when one thinks about manufacturability rather than idealized lab performance. That said, the limitations of PEO electrolytes are just as well known. Even after decades of work, ionic conductivity remains disappointingly low at moderate temperatures. Much of this traces back to the intrinsic crystallinity of PEO, which restricts segmental motion and, by extension, lithium transport. Also, interfaces formed between PEO electrolytes and high-energy electrodes are often unstable, both chemically and mechanically, which introduces additional resistance and accelerates degradation. These issues have proven stubborn, and incremental improvements alone have not been sufficient to push PEO systems into truly demanding battery configurations. One common response has been to introduce inorganic fillers, creating composite solid electrolytes (CSEs) that attempt to combine the best features of polymers and ceramics. Active fillers such as garnet-type lithium lanthanum zirconate (LLZO) are especially appealing because they offer fast lithium-ion conduction and excellent chemical stability. In theory, they should also reinforce the polymer matrix. In practice, however, the situation is more complicated. The interface between ceramic particles and polymer chains is rarely benign. High surface energy, poor chemical affinity, and mismatched mechanics often lead to filler aggregation and broken conduction pathways. Instead of helping lithium ions move more efficiently, the interface can become a dominant barrier. Among the different strategies explored, hydrogen bonding stands out because it is reversible, and tunable. Hydrogen bonds can disrupt PEO crystallinity and promote lithium salt dissociation, but they are not a universal solution. Their effects depend strongly on density and spatial arrangement. Too few bonds change very little; too many can lock up coordination sites and slow ion motion. The unresolved challenge, then, is not whether hydrogen bonding helps, but how to regulate it with enough precision to improve transport without creating new kinetic constraints. To this end, new research paper published in Materials Horizons and led by Fei Wang, Dr. Kai Chen, Xiaoxiao Li, Yan Fang, Mingjia Lu, Prof. Chao Zhang, Prof. Yue-E Miao and Prof. Tianxi Liu from the Donghua University, the researchers developed a PEO-based composite solid electrolyte featuring a precisely tunable hydrogen-bonded interfacial layer formed between amino-functionalized LLZO nanofibers and polymer chains. By optimizing hydrogen-bond density, they simultaneously enhanced polymer segmental mobility, lithium salt dissociation, and lithium-ion transference. This interfacial regulation produced continuous, low-barrier ion-transport pathways, leading to markedly improved conductivity and electrochemical stability.
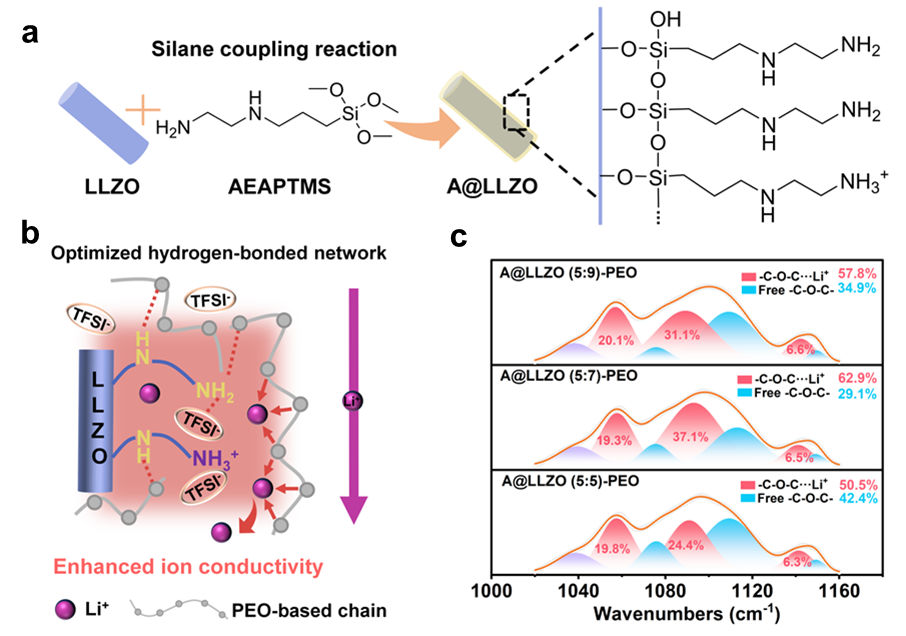
The research team modified LLZO nanofibers with an amino-functional silane coupling agent to generate well-defined organic–inorganic interfaces within a PEO-based electrolyte. And through controlled hydrolysis and grafting reactions, a series of surface-functionalized nanofibers with varying grafting densities were prepared and subsequently incorporated into polymer electrolytes by solution casting. Their approach enabled a direct comparison between insufficient, optimal, and excessive hydrogen-bond networks while holding the inorganic framework constant. The authors performed structural and spectroscopic analyses which showed that the silane modification formed a conformal organic layer on the LLZO surface without disturbing its crystalline garnet phase. More importantly, chemical signatures associated with amino and imino groups confirmed the presence of hydrogen-bond donors capable of interacting with both PEO ether oxygens and lithium salt anions. These interactions translated into markedly improved dispersion of the nanofibers within the polymer matrix, reducing aggregation and producing a more continuous ion-conduction landscape. Moreover, they conducted thermal and structural characterizations which showed that the introduction of hydrogen-bonded interfaces substantially suppressed PEO crystallinity. Furthermore, differential scanning calorimetry and diffraction analyses indicated an expansion of amorphous regions, consistent with enhanced chain mobility. However, the results also underscored a non-monotonic relationship between hydrogen-bond density and performance. At high grafting levels, excessive interfacial bonding competed with lithium ions for coordination sites on the polymer chains, partially negating the benefits of reduced crystallinity.
Additionally, they performed electrochemical measurements and found that ionic conductivity increased significantly when an optimal interfacial hydrogen-bond density was achieved, and reached values well above those of unmodified composites and pristine PEO. Simultaneously, the lithium-ion transference number rose, which indicates preferential lithium transport and reduced anion mobility. They also demonstrated using spectroscopic analysis that amino groups interacted with fluorinated anions, weakened lithium–anion associations and increased the population of free lithium ions available for conduction. The consequences of this interfacial regulation extended beyond bulk transport. Symmetric lithium cells assembled with the optimized electrolyte exhibited stable cycling over extended durations, accompanied by low polarization and delayed onset of short-circuiting. The authors post-cycling analyses reported the formation of mechanically robust, ionically conductive interphases enriched with lithium fluoride and lithium nitride species, both known to support uniform lithium deposition. When integrated into full lithium iron phosphate cells, the optimized composite electrolyte delivered sustained capacity retention and improved rate performance at elevated temperature.
In conclusion, the work of Donghua University scientists successfully established interfacial hydrogen-bond engineering as a genuinely powerful design principle for next-generation solid-state electrolytes. One of the most important findings to come out of this work is the realization that hydrogen bonds are inherently ambivalent and their impact depends, on how many are present and where they form. Indeed, what makes this contribution stand out is that it does not treat hydrogen bonding as a qualitative descriptor, but instead traces a clear boundary between regimes where these interactions accelerate lithium transport and regimes where they begin to work against it. Moreover, the authors elegantly offered a mechanistic picture that explains why performance improves and when it starts to degrade by systematically linking hydrogen-bond density to lithium coordination behavior, transference numbers, and activation energies. We believe equally important is the modularity of the strategy itself and the use of surface-functionalized ceramic nanofibers suggests a platform rather than a single solution. In principle, the same interfacial logic could be extended to other polymers, alternative functional groups, or different lithium salts. This opens realistic pathways toward operation at lower temperatures, compatibility with high-voltage cathodes, or improved long-term stability which are challenges that remain unresolved for many polymer-based systems. There are important implications for lithium metal anodes and by guiding lithium-ion flux more uniformly and stabilizing the solid electrolyte interphase, the engineered interface suppresses dendritic growth indirectly, without relying on excessive stiffness or thick barrier layers. That balance matters. It preserves the mechanical advantages of polymer electrolytes while addressing one of their most serious failure modes. More broadly, the new work in Materials Horizons reinforces a growing understanding in electrochemical materials: so-called “soft” interactions can exert surprisingly firm control over macroscopic battery behavior. Hydrogen bonds, often dismissed as weak or transient, are shown here to shape conductivity, cycling stability, and rate performance in a decisive way. In doing so, the study reframes interfacial design as central lever for advancing solid-state battery technologies.
Reference
Wang, Fei & Chen, Kai & Li, Xiaoxiao & Fang, Yan & Lu, Mingjia & Zhang, Chao & Miao, Yue-E & Liu, Tianxi. Precise Regulation of Hydrogen Bond Networks for Rapid Ion Transport in PEO-Based Composite Solid Electrolytes. Materials Horizons 2025, 12, 5872-5881.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.