Significance
The research team used publicly available 3D OCTA datasets encompassing the retinal region from the internal limiting membrane to the outer plexiform layer. Using an image projection network (IPN), they first segmented large and small vessels from en face projections, ensuring accurate labeling of arteries, veins, and capillaries. These segmented masks were then multiplied slice-wise across volumetric data to isolate corresponding vessel populations. For each subset, they computed transverse thickness—a voxel-based estimate of vessel diameter—used as a guiding constraint along the axial dimension. The iterative algorithm enforced depth-wise continuity consistent with physiological vessel thickness, effectively truncating artificial axial extensions lacking morphological coherence. The authors excluded low-intensity background regions through a threshold set at 10 % of the mean signal intensity in capillary zones to prevent spurious connectivity between unrelated vessels and the resulting artifact-suppressed images of large and small vessels were merged and further enhanced using Hessian filtering to restore natural rod-like geometries. They evaluated the quantitative performance using three established metrics: peak signal-to-noise ratio (PSNR), structural similarity index (SSIM), and mean-squared error (MSE) computed between consecutive B-scans and found in all comparisons, the proposed method outperformed both mean subtraction and conventional Hessian filtering, producing higher PSNR and SSIM and lower MSE, which jointly signify improved image fidelity. Moreover, visual analysis reinforced these metrics. In the en face and cross-sectional views, the new method removed deep-layer tails that persisted in other techniques while preserving the weak capillary network beneath major vessels. In intermediate vascular plexuses, tail artifacts that typically overlap with functional layers were effectively eliminated, revealing a smoother and anatomically consistent structure. Although a fraction of vessel signals entirely masked by severe artifacts remained unrecoverable—a limitation acknowledged by the authors—the reconstructed data displayed a remarkable reduction in redundant information. Furthermore, the authors’ quantitative morphometric characterization showed that after artifact suppression, distributions of vessel directional variance, waviness, and local coverage became narrower, reflecting more coherent spatial organization. Using canonical discriminant analysis across ten normal and ten DR samples, classification accuracy between the two groups rose from 65 % (cross-validated) in standard OCTA to 90 % after correction. They found the most distinctive improvement appeared in local coverage, strongly affected by tail artifacts, which directly influenced vessel-density quantification. Additionally, disease-related markers such as microaneurysms and intraretinal vascular abnormalities were more clearly delineated, enabling a physiologically grounded interpretation of diabetic microvascular remodeling. Indeed the new method delivered both computational and diagnostic gains which validated morphology-constrained filtering as an effective tool for 3D OCTA analysis.
In conclusion, the new work from Professor Zhiyi Liu and colleagues is an important advancement in OCTA image reconstruction with the development of new method integrating morphology-based filtering with transverse thickness characteristics to suppress axial tails in volumetric OCTA images. By linking physical vessel geometry to algorithmic filtering, they demonstrated that morphological self-consistency can guide artifact suppression more reliably than purely intensity-based corrections. The resulting images preserve the authentic 3D topology of retinal vasculature and minimize both false continuity and loss of fine capillary details. This sharpens visual interpretation and strengthens downstream quantitative analysis which is an important factor for automated disease assessment. Clinically, the implications extend beyond cleaner visualization. Tail artifacts have long hindered the use of 3D OCTA features in screening and longitudinal monitoring of retinal diseases. Their suppression opens the door to robust metrics of vascular integrity that may serve as biomarkers for early diabetic retinopathy, glaucoma, or ischemic pathologies. The reported 90% cross-validated classification accuracy underscores the diagnostic leverage achievable once redundant axial information is removed. Because the algorithm operates as a post-processing module, it can be integrated into existing imaging workflows without hardware modification and offer immediate translational potential. We believe equally important is the methodological generality. The approach relies on fundamental morphological cues—thickness and continuity—that are applicable to diverse tissue types imaged by OCT or other volumetric modalities. The authors envision combining this strategy with advanced acquisition schemes such as adaptive-gain OCTA (Ag1-OCTA), which modulates decorrelation times to balance signal preservation and artifact suppression. Future hybrid implementations may exploit reflectance-intensity cues or machine-learning-driven priors, forming a unified framework capable of multistage correction. The patent-pending status of the algorithm reflects its technical originality and expected clinical value. Indeed, their findings may catalyze renewed interest in post-acquisition correction pipelines, elevating OCTA from a qualitative tool to a quantitative instrument of precision ophthalmology.
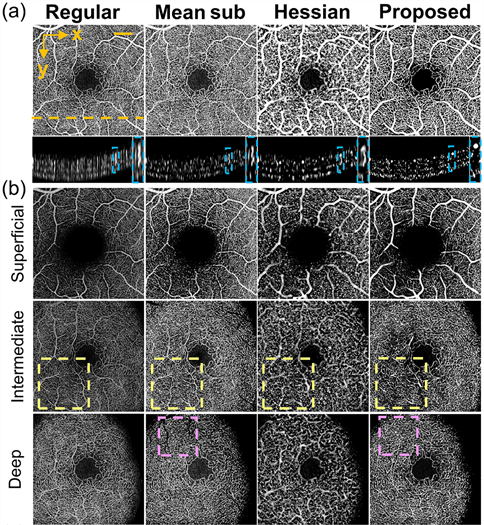
Comparison of tail artifact suppression using different methods.
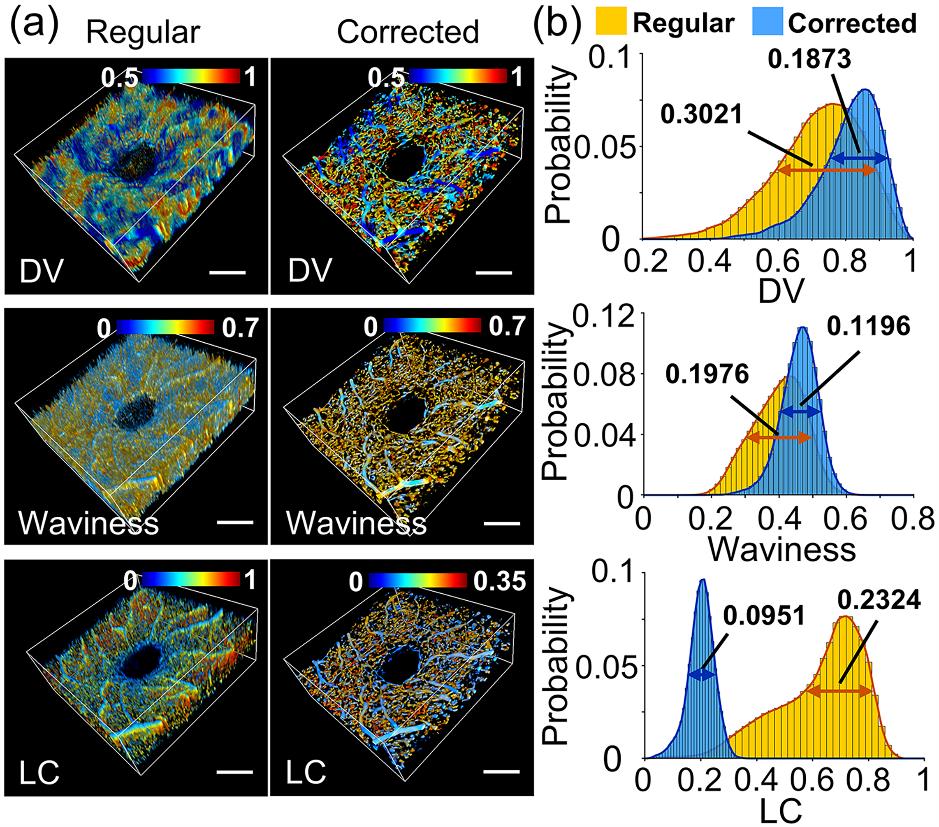
Comparison of quantitative characterization results of 3D OCTA before and after tail artifact removal.
REFERENCE
Lingxi Zhou, Jia Meng, Shuhao Qian, Zhihua Ding, and Zhiyi Liu, “Axial artifact suppression and precise characterization of three-dimensional optical coherence tomography microangiograms,” Opt. Lett. 50, 3986-3989 (2025)
Opt. Lett. Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



