Significance
Triple-negative breast cancer (TNBC) lacks expression of HER2, estrogen, and progesterone receptors. TNBC occurs in about 20–25 % of diagnosed breast cancers and is more likely to affect young people, African Americans, Hispanics, and those with BRCA1 mutation. Patients with TNBC do not benefit from endocrine therapy or trastuzumab since they lack appropriate targets for these drugs. Chemotherapy for TNBC has limited benefits and poor long-term survival rate. Additionally, TNBC is characteristically aggressive and has high recurrence rate. To develop novel and specific treatments and diagnostic tools for TNBC, new specific molecular agents are needed. Tumor-homing peptides are short peptides that localize specifically to tumor and tumor-associated microenvironment i.e., tumor vasculature, after systemic delivery.
Lymphatic metastasis are believed to be the most important spread pathways for many types of solid tumor and a key prognostic factor for survival of patients. Recently, the LyP-1 peptide isolated from the phagedisplayed peptide library has been recognized for its specific localization to tumors and their lymphatics. The LyP-1 peptide (CGNKRTRGC) is a circular peptide and has high specific binding and antitumor activity for cells carrying p32 protein on the cell surface, such as MDA-MB-435 melanoma cancer cells and pancreatic cancer BxPC-3 cells. The p32 gene is also highly upregulated in breast cancer cell lines such as MDA-MB-231 used in our studies.
Recently, it has been shown that LyP-1 binds specifically to p32 receptor on MDA-MB-231 cells. Following cancer diagnosis, it is crucial to evaluate cancer progression allowing monitoring of treatment efficiency, thus enabling potential treatment adjustments. Recent advances in imaging technologies such as magnetic resonance imaging (MRI) delivered a means of noninvasive quantification of biomarkers providing information on biologically relevant structure–function relationships in tumors. Functional molecular MRI imaging requires MR contrast agents that can selectively accumulate in the targeted triple-negative breast cancer and produce substantial change in MR contrast for anatomical images. Combining MRI capabilities with advanced contrast agents enables molecular profiling of target cells and the early detection of TNBC, thereby improving the accuracy of staging and treatment monitoring compared to routine MRI.
Magnetic iron-oxide nanoparticles (Fe3O4 NPs) are becoming an increasingly versatile and potent tool in modern medical imaging. They can be detected using standard MRI techniques due to their strong hypointense T2 signal. The chemical structure of MIO nanparticles also offers straightforward conjugation sites for tumor-specific biomolecules such as peptides to their biocompatible surface for tumor targeting. Reporting in Molecular Imaging and Biology, the researchers claim their work provides the groundwork for a systems nanotechnology approach to targeting that could lead to more sensitive location, diagnosis, and treatment of tissue- or cell-specific diseases. Their paper is titled “LyP-1 Conjugated Nanoparticles for Magnetic Resonance Imaging of Triple Negative Breast Cancer.” This was demonstrated by the researchers in a series of carefully designed experiments, according to the team.
In this study, we synthesized LyP-1 peptide conjugated with iron oxide NPs to create a specific TNBC-targeted MR contrast agent for tumor diagnosis and monitoring. In this study, we demonstrate the effectiveness of Fe3O4NPs conjugated with LyP-1 peptide as a TNBC-targeting MR contrast agent. The estrogen receptor antagonist (Tamoxifen) and anti-HER-2 antibodies (Trastuzumab and Pertuzumab) have been used clinically in targeted therapies for hormone and HER-2 receptor-positive breast cancers. Unfortunately, currently, there is no triple-negative breast cancer (TNBC)- targeted treatments available, thus there is a need for personalized therapy.
Early stages of TNBC can be diagnosed using immunohistochemical and fluorescence in situ hybridization analyses based on the negative expression of estrogen and progesterone receptors and HER-2 in excision biopsy samples of breast cancer. Imaging modalities such as mammograms, MRI, and ultrasound usually detect TNBCs at much later stages of the disease when tumor lesions are larger than 2.5 cm. Development of advanced molecular imaging techniques should provide informed therapeutic monitoring and better treatment for drug-resistant cancer cells which will result in prognostic improvement of TNBC patients. There are several potential molecular biomarkers of TNBC based on over-expression of epidermal growth factor receptor, insulin growth factor 1 receptor, mucin 1, CD44, folate receptor, and CXCR4. However, because umor stroma is a main barrier for delivering molecular contrast agents, nanoparticle-based targeted contrast agents to tumor cell surface can only be delivered into the tumor interstitial space by Bleaky^ tumor vasculatures mediated by the EPR effect. To overcome this barrier, in our studies, we targeted the tumor vessels surface using LyP-1 homing peptide. As TNBC resists current therapy, there is a need for early detection of TNBC to improve outcome. Real-time and noninvasive molecular imaging provides this capability; that is not available with currently used immunohistochemistry and fluorescence tests in situ.
LyP-1 was originally identified as a tumor-homing peptide that specifically recognizes tumor cells, tumor lymphatics, and tumor-associated macrophages. Significant p32 expression which is thought to be the receptor for LyP-1 was common in human cancers including breast cancer [22]. Recently, LyP-1 peptide has been used successfully to synthesize a bispecific antibody conjugated with doxorubicin and LyP-1 which showed cytoxicity in MDA-MB-231 breast cancer cells in vitro.
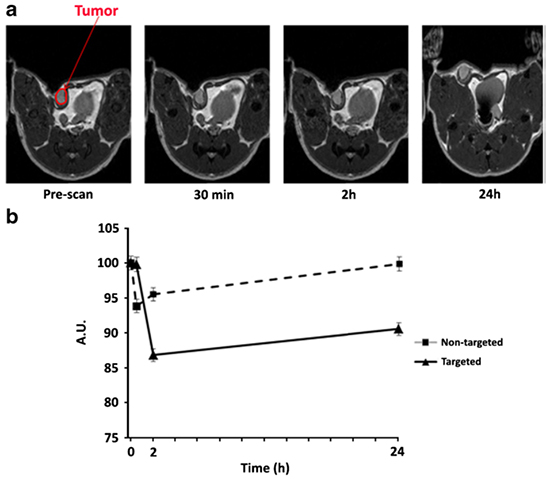
In this study, we utilized the LyP-1 peptide for early detection of breast cancer in vivo using MRI, explains Dr. Abedelnasser Abulrob, the senior research officer at the NRC Canada and professor at the Faculty of Medicine-University of Ottawa, who led the study. Dr Abulrob then added “when we injected the molecular imaging agent [LyP-1]-Fe3O4-Cy5.5, we observed significant tumor retention of [LyP-1]-Fe3O4- Cy5.5 in the TNBC tumor foci. The imaging agent saturated the tumor and its surrounding sites within 2 h after injection”.
In a nutshell Dr. Abulrob and his colleagues developed [LyP-1]-Fe3O4-Cy5.5 that can recognize TNBC as the MR contrast agent. The [LyP-1]- Fe3O4-Cy5.5 revealed TNBC-specific binding to TNBC tumor cells and enabled TNBC-specific MR imaging of tumors in vivo. The early MR imaging of TNBC using LyP- 1-Fe3O4 could assist in both pre-treatment planning and the prognosis, as well as adding to our understanding of the biological behavior of TNBC. The authors suggest that [LyP-1]-Fe3O4 may serve as a robust MR contrast agent for diagnosis of TNBC in humans.
Reference
Abedelnasser Abulrob, Slavisa Corluka, Barbara Blasiak, Gino Fallone , Dragana Ponjevic, John Matyas , Boguslaw Tomanek. LyP-1 Conjugated Nanoparticles for Magnetic Resonance Imaging of Triple Negative Breast Cancer. Molecular Imaging and Biology 2018 Jun;20(3):428-435. doi: 10.1007/s11307-017-1140-4.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


