Significance Statement
Nitroaromatics have been considered the most common organic pollutants in industrial wastes that are discharged. They have been widely implemented in industrial processes such as in the manufacture of dyes, pharmaceuticals, wood preservatives, rubber chemicals, pesticides, and pigments. However, nitroaromatics pose threat to human life due to their carcinogenicity that may leave residues in animal products such as eggs and meat. They therefore have been listed as the top 114 organic pollutants in the diverse nitroaromatic compounds and are a threat to human health owing to bioaccumulation, high toxicity, a difficulty in biochemical degradation of carcinogenesis, mutagenesis, and terotogenesis.
Luckily, the reduced forms of the nitrophenols, aminophenols, have considerably low toxicity. They also serve as intermediates in the manufacture of dyestuffs, antipyretic drugs, and other fine chemicals. The synthesis of the aminophenols is normally accomplished via the typical catalytic reduction of nitrophenols in the presence of noble metals or strong iron-acid catalysts. Unfortunately, during these reactions, metal oxides are inevitably formed as by-products of iron-acid catalyst, as lowers the catalytic efficiency and restricts the use of metallic reagents. On the other hand, though noble metals have high catalytic efficiency, high hydrogen pressure and elevated temperatures are critical prerequisites in the reaction.
For this reason, obtaining aminophenols through catalytic hydrogenation is a promising path owing to milder reaction, high cost-effectiveness, and high conversion efficiency. Therefore, it is important to find a green and high efficient catalyst for the reduction of 4-nitrophenol to 4-aminophenol.
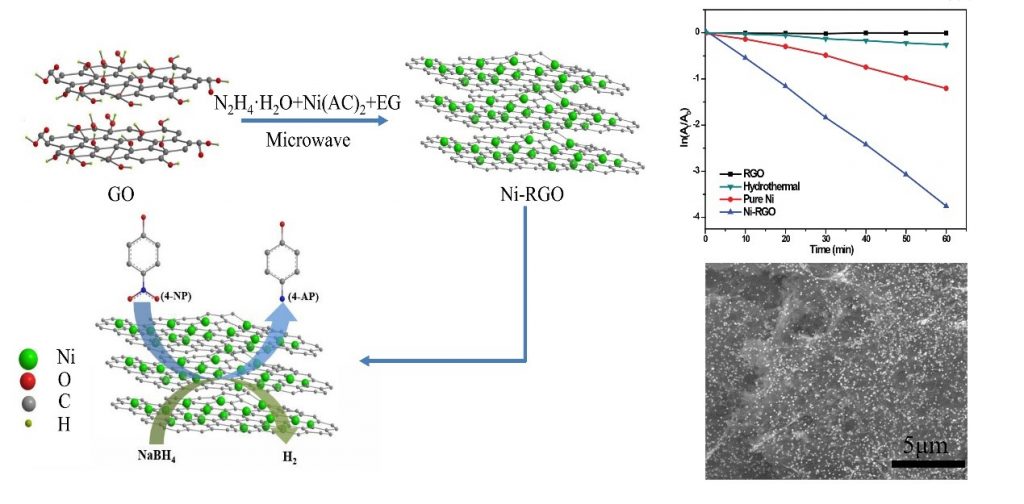
Researchers led by Professors Hanxun Qiu at the University of Shanghai for Science and Technology in China developed a facile route for the synthesis of nickel nanoparticle decorated reduced graphene oxide nanocomposites in a microwave-involved process. They also demonstrated the composites’ excellent catalytic performance. In their research, glycol was subtly used as both a solvent and a reducing agent. Their research work is published in Applied Surface Science.
The authors prepared nickel nanoparticle decorated reduced graphene oxide nanocomposites with excellent catalytic performance in the reduction of 4-nitrophenol though an environmentally friendly microwave assisted method. They systematically characterized the morphology as well as structure of the as-prepared samples.
The researchers evaluated the catalytic performance through the time-dependent UV-vis absorption spectra. This evaluation indicated that the reduction of the 4-nitrophenol to 4-aminophenol was successfully realized in the presence of the as-prepared nickel nanoparticle decorated reduced graphene oxide nanocomposite.
The results of the study indicated that nickel nanoparticles with an average diameter of 40nm were homogeneously anchored onto the surface of the reduced graphene oxide sheets. The pseudo-first-order kinetic reaction constant demonstrated that the prepared nanocomposite with over 64wt% loading nickel nanoparticles possess superior catalytic activities and selectivity toward the reduction of 4-nitrophenol compared to the bare nickel nanoparticles as well as reduced graphene oxide sheets. The corresponding kinetic reaction rate constant was even 4-fold over the bare nickel nanoparticles. Such promising nanocomposites exhibit great potential for the green treatment of industrial effluent containing nitrophenol in an elementary, green and sustainable approach.
Reference
Hanxun Qiu, Feilong Qiu, Xuebin Han, Jing Li, Junhe Yang. Microwave-irradiated preparation of reduced graphene oxide-Ni nanostructures and their enhanced performance for catalytic reduction of 4-nitrophenol. Applied Surface Science, volume 407 (2017), pages 509–517.
Go To Applied Surface Science
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


