Significance
Hydrogen has steadily gained traction as the alternative clean for fossil fuel. Among the technologies built around hydrogen, high-temperature proton exchange membrane fuel cells—HT-PEMFCs—stand out because they are efficient, tolerate contaminants like CO better than their low-temperature counterparts, and don’t require complex water management systems. These systems operate at elevated temperatures, typically between 120 and 200 °C, and rely heavily on the performance of a key component: the proton exchange membrane (PEM). It’s not just a passive barrier—it’s where proton conduction happens, and the entire system’s efficiency hinges on it. But the membrane also has to survive long-term exposure to heat, acid, and mechanical stress without falling apart. That’s a tough job for any material. Right now, phosphoric acid-doped polybenzimidazole (PBI) membranes are among the best options available for HT-PEMs. Especially the meta-structured versions like mPBI, which have been widely studied and used commercially. They’ve proven to be thermally and chemically stable, which is essential at the temperatures fuel cells run. But these materials aren’t without problems. In fact, they’re kind of a nightmare to process. They don’t dissolve well in common solvents, making them hard to work with when it comes to membrane fabrication. Their rigid backbone structure also means they’re brittle, particularly after being doped with phosphoric acid to enhance conductivity. And while high acid doping is necessary to get the performance up, it introduces other issues—acid leaching, catalyst degradation, and performance loss over time. A lot of effort has gone into modifying the PBI backbone—adding side chains, crosslinking, blending with other polymers—to try and make it more flexible or improve acid retention. But often these fixes are a trade-off. You might gain in processability but lose conductivity, or improve doping levels but compromise stability. It’s been difficult to find an approach that actually improves everything at once.
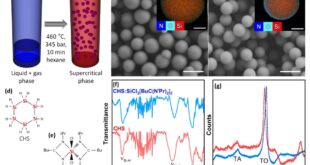
To this account, new research paper published in International Journal of Hydrogen Energy and led by Professor Shoulei Miao from the College of Chemical Engineering and Material at Heilongjiang University and conducted by Yamei You, Xinyang Deng, Qian Liu, and Professor Yanjun Hou, the researchers synthesized and characterized a novel series of semi-flexible PBI materials. Initially, the goal was to prepare the poly(5-phenyl-1H-1,2,3-triazole) or QP unique monomer, using Suzuki coupling reactions. Once synthesized, they integrated varying amounts of QP into the rigid PBI backbone through controlled melt polycondensation, and produced new block copolymers which they named PBI-QP-15 and PBI-QP-20. The numerical suffixes (15 and 20) denote the percentage of QP incorporated into each polymer chain. The researchers used nuclear magnetic resonance spectroscopy (¹H NMR) to verify the successful synthesis of these copolymers and observed distinct signals corresponding to both imidazole rings and newly introduced QP units. The integrated peak areas scaled proportionally with increasing QP content, enabling precise quantification. These results were also supported by FTIR spectroscopy where characteristic absorption bands clearly confirmed the presence of both imidazole and triazole rings. Further validation came through X-ray photoelectron spectroscopy, which provided insights into the polymer’s chemical bonding environment. Notably, analysis of the C1s and N1s spectra revealed shifts consistent with increased C–C bonding and decreased C=N groups, indicative of successful QP incorporation altering the electronic environment of the polymer chains. The next critical assessment was how these chemical modifications translated into physical and practical properties. Addressing solubility—historically problematic with conventional PBI—the team discovered that both PBI-QP-15 and PBI-QP-20 readily dissolved at room temperature in common solvents such as DMF, DMSO, and formic acid and by this offer much simpler processing compared to conventional PBIs which typically demand harsh conditions and strong solvents. Membranes fabricated from these materials underwent rigorous microstructural analysis. Scanning electron microscopy revealed exceptionally smooth and uniform surfaces and cross-sections, free from aggregates or phase separation. Atomic force microscopy, however, showed distinct microphase separation—a desirable trait here—as it created interconnected proton-conducting channels. This separation likely arises from hydrophilic imidazole clusters surrounded by hydrophobic phenyl regions, balancing proton conductivity with structural integrity.
To validate practical performance, the authors investigated the membranes’ phosphoric acid uptake capacity—a critical factor for HT-PEMFCs. Impressively, PBI-QP-20 demonstrated an acid doping level (ADL) of 10.5, nearly doubling the 5.3 ADL of traditional PBI. Contact angle measurements confirmed increasing hydrophilicity correlating with greater QP content, helping the membranes effectively retain absorbed acid. Crucially, dimensional stability remained robust, attributed to the rigidity imparted by benzene rings within the QP units. Evaluating thermal stability, thermogravimetric analysis revealed that PBI-QP-20 maintained over 72% of its mass even at temperatures reaching 800°C, surpassing conventional PBI. Oxidative stability, tested under harsh Fenton’s reagent conditions, also showed that PBI-QP variants degraded significantly slower, likely due to their semi-flexible structure effectively shielding vulnerable sites from radical attack. Finally, proton conductivity tests under anhydrous conditions yielded encouraging results. The authors noticed that PBI-QP-20 membrane achieved a peak conductivity of 64.3 mS/cm at 170°C—significantly higher than standard PBI membranes. Long-term stability was also strong, with over 67% acid retention after 108 hours at 80°C and moderate humidity, coupled with minimal loss in conductivity. The authors subjected them to real-world fuel cell tests and highlighted the membrane’s promise and when integrated into membrane electrode assemblies and tested in hydrogen/oxygen fuel cells without humidification or backpressure, PBI-QP-20 exhibited a peak power density of 573.6 mW/cm² at 180°C. Additionally, the open circuit voltage remained stable around 1.0 V, highlighting excellent gas impermeability and electrochemical stability. These results mark a significant advancement toward scalable, efficient, and reliable membrane materials for next-generation fuel cell technologies.
In conclusion, the research work of Professor Shoulei Miao and colleagues has immediate and practical implications in the facilitation of scalable membrane manufacturing. The newfound solubility of PBI-QP polymers in widely used solvents at ambient temperature removes a significant bottleneck in industrial processing. Traditional PBI typically requires harsh solvents and elevated temperatures to become processable, making it costly and complex to fabricate. In contrast, these new materials can be cast into membranes with greater ease and consistency, all while lowering energy demands—a major step forward for commercial viability. Moreover, the superior acid doping levels and proton conductivity observed in PBI-QP-20 are especially promising for HT-PEMFCs. Applications such as combined heat and power systems or heavy-duty vehicles, which demand high performance under dry and elevated temperature conditions, could particularly benefit. With its enhanced conductivity and acid retention, PBI-QP-20 suggests a membrane that can deliver sustained output over extended periods, reducing maintenance intervals and increasing system reliability. In contexts where devices must endure thousands of hours of continuous operation under thermal stress, such improvements in durability are highly consequential. Equally noteworthy is the innovative use of semi-flexibility as a deliberate design principle. Instead of forcing pliability into a rigid backbone or compromising structural stability by adding flexible side chains, the team achieved a delicate balance. The QP segments introduced enough molecular freedom to ease internal stresses, improve acid absorption, and support more effective proton transfer—without undermining the integrity of the overall polymer network. This kind of architectural nuance illustrates how tailoring molecular conformation can translate into tangible performance advantages at the device level.
Reference
Yamei You, Xinyang Deng, Qian Liu, Yanjun Hou, Shoulei Miao, A semi-flexible polybenzimidazole with enhanced comprehensive performance for high-temperature proton exchange membrane fuel cells, International Journal of Hydrogen Energy, Volume 78, 2024, Pages 879-888,
Go to Journal of Hydrogen Energy. Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.