Significance
Several neurodegenerative diseases such as Alzheimer’s disease, Creutzfeldt–Jakob disease and Prion disease are caused by protein misfolding. For example, the aggregation of amyloid-beta peptides into fibrils as well as the accumulation of fibrils into the senile plaques in the extracellular space in the human brain is a footprint of the Alzheimer’s disease. Although plaques have been found to occur in the extracellular space of the brain, there exists an evidence that amyloid-beta processing, aggregation, and assembly into soluble oligomers could occur inside the brain cells. In fact, it has been found that oligomers in the form of trimers and dimers are more neurotoxic than the fibrillar assemblages.
Unfortunately, the oligomers are transient in nature, making it difficult to characterize them by the traditional methods. This suggest that advanced methods should be implemented. Researchers led by Professor Yuri Lyubchenko at the University of Nebraska Medical Center developed a number of single-molecule techniques such as tethered approach for probing intermolecular interactions using AFM-based single-molecule force spectroscopy that have the capacity to probe dimers of the amyloid-beta peptides.
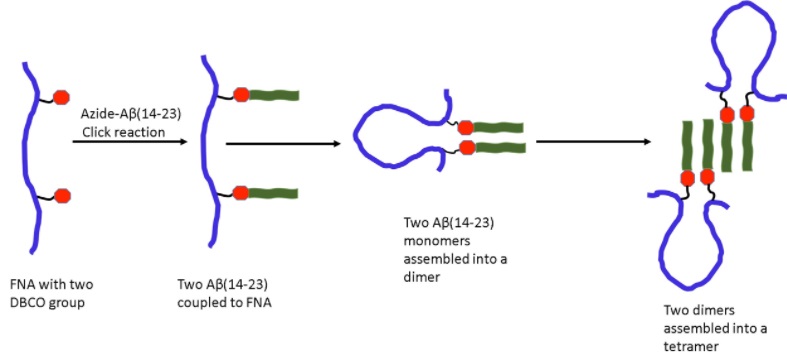
The same team has recently developed an applied a novel polymer nanoarray method for probing tetramers and trimers formed by the amyloid-beta (14-23) segment of an amyloid-beta protein at the single-molecule level. By implementing this method in conjunction with the tethered approach for probing intermolecular interactions and AFM-based force spectroscopy, the authors were able to characterize the effect of pH on the aggregation of these oligomers. Their research work is published in journal, Physical Chemistry Chemical Physics.
Although their current study provides a strong support for the implementation of single-molecule biophysics methods, in order to characterize amyloid oligomers, additional methods are necessary for probing higher order structures generated by the same monomeric units. Flexible nanoarray is one of these methods where the same two peptides are end-tethered inside of a flexible polymer allowing them to interact and create a dimer. This idea has been adopted in this study in order to probe tetramer and trimer.
The authors found from the experimental results that pH affected the oligomer assembly process. At a pH of 7, tetramers and trimers agglomerated into structures with an identical stability, while at an acidic pH, the oligomers adopted a group of structures with varying lifetimes and strengths.
The research team also realized that the dimers were stabilized by strong interactions between monomers with lifetimes in the second timescale. This fact was supported by measuring lifetimes directly using the single-molecule fluorescence tethered method for probing of intermolecular interactions. The amyloid-beta (14-23) segments of both Amyloid -beta 40 and amyloid beta-42 proteins have a critical role in the fibril assembly process by stabilizing these fibrils via the formation of an extended Beta-sheet structure. The study indicated that the interaction between the amyloid beta (14-23) monomers with the hairpin was stronger as compared to the interaction between two monomers. These outcomes suggested that the hair pin structure contributed to oligomer stability.
The major finding of their study suggests that oligomeric species of identical size can take a string of structures having different stability. This structural heterogeneity implies that the amyloid self-assembly process is complex and could take varying pathways. The study by Yuri Lyubchenko and co-workers contributes to further understanding of protein misfolding which will ultimately help finding a better treatment for serious neurodegenerative diseases.
Reference
Sibaprasad Maity, Ekaterina Viazovkina,b Alexander Gallb and Yuri L. Lyubchenko. Single-molecule probing of amyloid nano-ensembles using the polymer nanoarray approach. Physical Chemistry Chemical Physics, volume 19 (2017), pages 16387—16394.
Go To Physical Chemistry Chemical Physics
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

