Significance
Biocompatible scaffolds are commonly used in tissue engineering to realize multiple synergistic functions for regeneration owing to their ability to provide structural tissue support and facilitate cell adhesion and mass transport. It is well-known that neural tissues can react to stimuli. Taking advantage of this, biochemical and biophysical stimulation has attracted attention as an additional strategy for promoting functional regeneration and improving neurite growth. Neural stimulation techniques, specifically electrical stimulation, have been widely investigated to promote neural regeneration by modulating neuronal activities.
While it is possible to apply an electrical current or field through ionically conductive tissue fluid, electrically conductive neural scaffolds are required for more controllable and efficient delivery. Such scaffolds have effectively facilitated nerve regeneration, including carbon-based materials and conductive polymers. Nevertheless, delivering electrical stimulation to conductive scaffolds in clinical applications remains a big challenge. While most current solutions are limited to transcutaneous stimulation after surgery, others require transcutaneous wires that increase infection risks. Although self-powered and wireless electronic systems have emerged as promising solutions, they may cause undesirable impacts due to the spread of the electric current beyond the injury site.
Lately, photoacoustic stimulation has been identified as a promising high-precision and nongenetic neural stimulation method. The process involves generating acoustic waves at ultrasonic frequencies by delivering pulsed light into the absorbers. As a result, it has been speculated that introducing efficient absorbers like carbon nanotubes (CNTs) into biocompatible materials could induce photoacoustic stimulation in these biomaterials for use as scaffolds. Neural scaffolds with photoacoustic stimulation could replace optogenetics and electrical stimulation as they can promote neurite outgrowth without wired connections or viral transfection.
Herein, researchers from Boston University: PhD candidate Nan Zheng, Ran Cheng, Dr. Linli Shi and Professor Chen Yang in collaboration with Dr. Vincent Fitzpatrick and Professor David Kaplan from Tufts University designed a generalized strategy for integrating photoacoustic neural stimulation into hydrogel scaffolds via nanocomposite approach. Polyethylene glycol (PEG)-functionalized CNTs were embedded into silk fibroin to form soft and biocompatible photoacoustic scaffolds. Their work is currently published in the journal, ACS Nano.
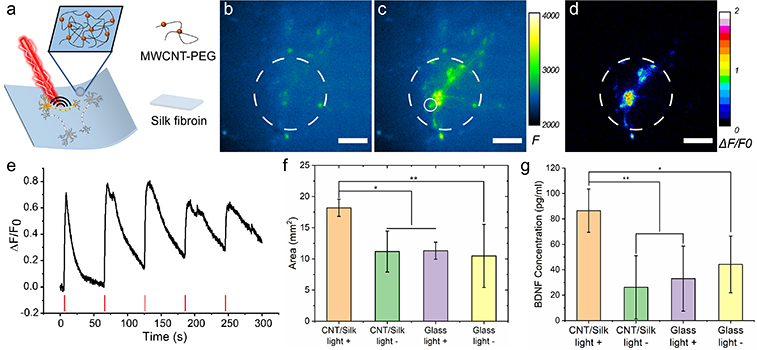
The authors showed that the biocompatible silk scaffold with photoacoustic capabilities promoted neuronal regeneration by enabling nongenetic activation with spatial precision. Upon excitation, the CNT/silk film generated a broadband photoacoustic wave, whose amplitude could be controlled by varying the laser energy and CNT concentration. For successful neural stimulation, the generated photoacoustic wave required pressure of about 0.185 MPa. The CNT/silk films supported neural growth and had no impact on the viability or morphology of the film.
The biocompatibility, reliability and repeatability of the neural stimulation function of the scaffold were confirmed through time-resolved calcium imaging, activating over 94% of the photoacoustic-stimulated neurons in the light illuminated area. Compared with the unstimulated control group, the scaffold was observed to promote neurite outgrowth by 1.74-fold, which was also associated with an increase in the brain-derived neurotrophic factor concentration.
Compared with the existing neural stimulation methods, this is a light-mediated method with a longer wavelength and sufficient penetration depth in tissues and can potentially provide noninvasive postoperative stimulation. Moreover, it does not require genetic transfection, which has been associated with numerous technical and ethical issues, and avoids the difficulty of controlling injected nanoparticles by allowing the nanoparticle transducers to function at the implanted sites. The findings confirmed the possible involvement of transient membrane disruption and calcium ion activation channels and the reasons behind the long decay constant.
The repair of the nervous system is one of the great challenges of modern medicine. Although significant advances have been made in cell-based therapies, nanotechnologies, and novel biomaterials, as well as a better understanding of the nervous system’s knowledge, however, tissue engineering advocates the combination of innovative biocompatible scaffold like the one reported by Nan Zhang and colleagues together with cells and bioactive molecules to mimic perfectly the environment to regenerate while supporting the repair of new tissue. Of course, more multidisciplinary approaches and research will be needed in the future to apply these therapeutic strategies in clinics.
In summary, a photoacoustic CNTs embedded silk scaffold was reported for application in light triggered neural stimulation and regeneration. The study provided more insights into the action mechanism of the emerging photoacoustic neural stimulation method, providing clarifications and answers to some speculations and hypotheses. Owing to their advantages and design flexibility, as well as the efficacy of photoacoustic stimulation, the authors believe that the photoacoustic CNT/silk scaffolds provide an additional and more robust method for future generation nonpharmacological neural regeneration.
Reference
Zheng, N., Fitzpatrick, V., Cheng, R., Shi, L., Kaplan, D., & Yang, C. (2022). Photoacoustic Carbon Nanotubes Embedded Silk Scaffolds for Neural Stimulation and Regeneration. ACS Nano, 16(2), 2292-2305.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



