Significance
Lithium metal is widely used as an anode for solid-state batteries because of its excellent capacity and the fact that the potential is as low as it gets. However, in practice, performance is usually dictated by something much less ideal: loss of contact with the solid electrolyte. In liquid cells, the electrolyte can flow and partly absorb the volume changes that come with stripping. That option disappears once both phases are solid. Here, lithium transport depends on the interface staying intact, both physically and chemically. As stripping proceeds, vacancies are injected at the metal surface. They don’t disappear on their own. If diffusion from the bulk can’t keep pace, those vacancies accumulate, and the interface drifts into an unstable state. Voids then begin to form. Slowly at first. Later they grow, coalesce, and reshape the current distribution. Local current density rises where contact remains, which only accelerates the same processes that caused the problem in the first place. Failure doesn’t happen abruptly, and it is the endpoint of a sequence that is already underway long before the voltage spikes and attempts to mitigate this behavior have generally fallen into two categories. One relies on external control. Applying stack pressure or increasing temperature can promote lithium creep and restore contact, at least temporarily, but these measures come with trade-offs. Cell architectures become more complex, and mechanical stresses increase, which is especially problematic for stiff or brittle electrolytes. The other category focuses on modifying the electrode itself. Interfacial coatings, alloy layers, or engineered interphases are intended to adjust wetting behavior or local reaction kinetics. Many of these ideas work well in early cycles. Over longer stripping periods, though, their advantage often fades, which hints that the underlying processes driving void formation are only being delayed, not addressed.
Lithium–magnesium alloys sit somewhere in between these strategies. Even small amounts of magnesium change the picture. Magnesium enters lithium as a solid solution, raising surface energy and altering how lithium atoms move through the metal, without adding a second active phase. Experiments consistently show smoother deposition and better contact retention. At the same time, there is a cost. As magnesium content increases, lithium diffusion slows, and usable capacity drops when current demand is high. That trade-off is hard to ignore. Stabilizing the interface appears to come at the expense of transport within the electrode. What is still lacking is a framework that treats vacancy generation, diffusion, void nucleation, and interfacial breakdown as parts of the same problem. Many models focus on one mechanism at a time. Useful, but incomplete. Without capturing how these processes interact at intermediate length scales, it remains difficult to predict when alloying helps, when it hurts, and why. Recent research paper published in Chemistry of Materials and conducted by Ke Li, Jundi Huang, Xinyi Qu, Gaoming Fu, Xiang Chen, Weijia Shen and led by Professor Yixin Lin from the School of Energy and Power Engineering at the Huazhong University of Science and Technology, the researchers developed a novel coupled electrochemical–diffusion–nucleation–phase-field model to simulate stripping-induced interface evolution in lithium and lithium–magnesium anodes. The framework explicitly links vacancy generation, stochastic void nucleation, and lithium transport within a single computational scheme. It also distinguishes physical contact failure from diffusion-limited chemical failure as magnesium content increases. Plus, the new approach enables composition- and current-dependent evaluation of stripping capacity without presupposing interfacial defects.
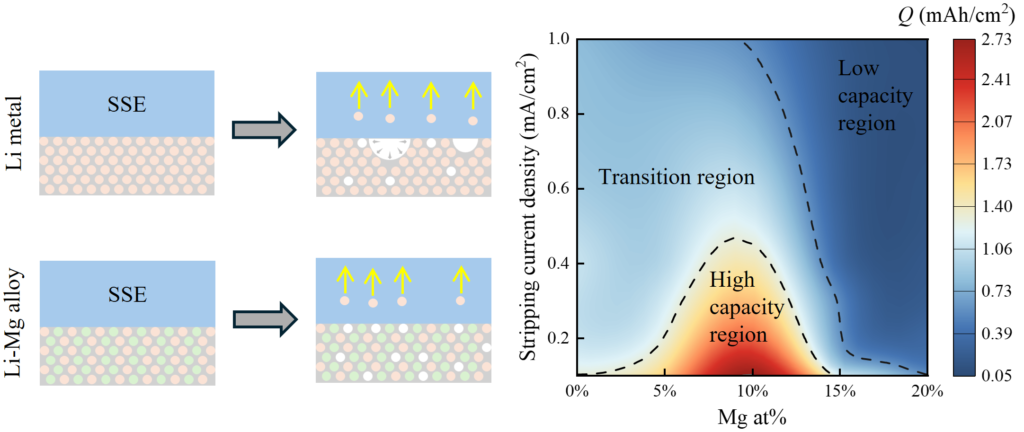
The research team constructed a coupled electrochemical–diffusion–nucleation–phase-field framework to track how vacancies accumulate, voids nucleate, and interfaces degrade during stripping. The investigators began by examining pure lithium anodes under different stripping current densities, allowing the model to evolve from an initially ideal, fully contacted interface. At low current density, the authors observed an extended incubation period during which vacancies accumulated without immediate void formation. Once nucleation occurred, voids deepened through combined lateral expansion and vertical growth, producing a thick interfacial void layer before failure. Higher current density shortened this incubation stage and shifted growth toward shallow, laterally connected voids, leading to earlier loss of contact despite a thinner void layer. The team then examined lithium-magnesium alloys across a range of magnesium contents. The researchers found that even modest magnesium incorporation delayed void nucleation and preserved interfacial contact over a larger fraction of the stripping process, and also they found magnesium content increased from low to intermediate levels, the stripping capacity required to initiate void formation rose, and the resulting void layers became thinner and less disruptive. At these compositions, voltage increases occurred without immediate contact loss, indicating that transport limitations rather than physical separation governed failure.
At higher magnesium content, the authors observed a distinct shift in failure mode. Void formation became energetically unfavorable, and the interface remained intact throughout stripping. Failure instead arose from lithium depletion near the interface, as diffusion through the alloy couldn’t sustain the imposed current. The investigators linked this behavior to the strong dependence of lithium diffusivity on magnesium concentration, which limited the depth of the electrode participating in bulk stripping. By separating total stripping capacity into surface-mediated and bulk-diffusion-driven contributions, the study demonstrated a gradual transition from surface-dominated stripping in pure lithium to bulk-controlled behavior in magnesium-rich alloys. The researchers further explored how current density modulated these effects, showing that alloys outperform pure lithium at low current density but lose that advantage as current increases. Repeated stripping with intervening rest periods revealed that alloys retaining interfacial contact could recover partial capacity through lithium redistribution, but pure lithium anodes can’t.
In conclusion, the work of Professor Yixin Lin and colleagues explains why stripping behavior varies not only with composition but also with current density and time by treating void nucleation as a stochastic but energetically governed process. Their new findings clarify also why lithium-magnesium alloys stabilize interfaces under certain conditions but underperform under others, resolving inconsistencies across prior experimental reports. The results also highlight that interfacial stability alone doesn’t guarantee usable capacity. Suppressing voids without maintaining sufficient lithium transport simply shifts the failure mechanism from physical separation to chemical starvation. This distinction matters for anode design, particularly in systems operating without external pressure, where diffusion limitations become more pronounced. The interdependence diagram developed in the study provides a structured way to think about this trade-off, identifying regimes where alloying improves performance and others where it becomes counterproductive.
On top of that, we believe the new modeling strategy demonstrates how mesoscale approaches can connect atomistic properties, such as surface energy and diffusion coefficients, to macroscopic battery behavior. While the conclusions are bounded by the assumptions of pressure-free operation and idealized interfaces, the framework provides a foundation for incorporating additional effects such as mechanical constraint or creep. And in that sense, the work doesn’t prescribe a single optimal alloy composition but rather establishes criteria for matching material choice to operating conditions.
Reference
Li, Ke & Huang, Jundi & Qu, Xinyi & Fu, Gaoming & Chen, Xiang & Shen, Weijia & Lin, Yixin. (2025). Enhancing Lithium Metal/Solid-State Electrolyte Interface Stability by Magnesium Alloying: Void Nucleation, Growth, and Interfacial Failure. Chemistry of Materials. 37. 5884-5898. 10.1021/acs.chemmater.5c01122.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.