Through Indium (III) Chloride Trimethylsilyl Chloride or Scandium (III) Triflate Catalysis
Significance
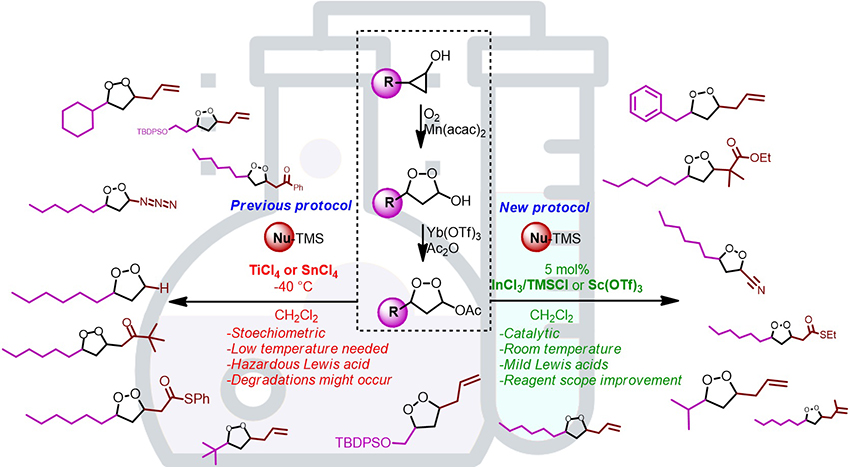
Malaria is a disease caused by a plasmodium parasite transmitted by the bite of infected mosquitoes. Symptoms include: chills, fever and sweating, and the disease can at times be fatal. Therefore, intensive research have for decades been channeled towards developing a drug to treat and even prevent the disease, for instance, Artemisinin or Arterolane antimalarial drugs that comprise of endoperoxides. Endoperoxides are a rich source of natural or unnatural bioactive products, leading to the development of important anti-malarial drugs, however some compounds were also known to possess anti-bacterial, anti-fungal, anti-viral or cytotoxic activities. In a recent publication, researchers demonstrated a methodology to build disubstituted 1,2-dioxolanes through Lewis acid activation of 1,2-dioxolanyl acetates with Titanium tetrachloride (TiCl4) or Tin (IV) chloride (SnCl4) at low temperature. The methodology was still difficult to control since a slight modification of the Lewis acid stoichiometry was reported to lead to more degradation products (if in excess) or a worse conversion in the desired product (if in deficiency), thus making the experiments difficult to reproduce at times.
Therefore, an alternative approach utilizing a mild Lewis acid under catalytic amount at room temperature would be highly desirable to circumvent the degradation products and the utilization of sub-stoichiometric amount of Lewis acid under low temperature conditions. With the goal being to sidestep the aforementioned challenges, researchers from the Paris-Saclay University in France: Alexis Pinet, Dr. Bruno Figadère and Dr. Laurent Ferrié, developed a novel catalytic protocol of functionalization for 1,2-dioxolanyl acetates, identifying two efficient systems, namely Indium(III) chloride/ Trimethylsilyl Chloride and Scandium (III) Triflate. Their work is currently published in the research journal, Advanced Synthesis & Catalysis.
The researchers prepared some endoperoxyhemiacetals with oxygen and manganese salts by ring expansion from readily available cyclopropanols, then the acetoxy function was introduced by Lewis acid catalyzed acetylation, which was crucial for such compound. The functionalization of 1,2-dioxolanes was then undertook at room temperature under catalytic conditions. During these approaches both Indium (III) Chloride/Trimethylsilyl Chloride and Scandium (III) Triflate were used for purposes of evaluating their catalytic properties.
The authors reported the two catalysts allowed milder and more practical conditions than those previously reported with improvements in scope and reproducibility. In fact, their approach was reported to allow a full catalytic sequence from cyclopropanols to produce desired functionalized 1,2-dioxolanes.
In summary, the study demonstrated the use of catalytic amount of scandium (III) triflate or indium (III) chloride (with trimethylsilyl chloride) for the functionalization of endoperoxyacetals through Sakurai or Mukaiyama reactions. In a statement to Advances in Engineering, Dr. Laurent Ferrié mentioned that, although some poorly reactive species such as deactivated allylsilanes, and hindered nucleophiles work better with Tin (IV) chloride and Titanium tetrachloride, the two new catalytic systems enlarged the reagents scope to ester and thioester enol ether derivatives, which made complementary the catalytic and the stoichiometric versions of the studied transformation.
Reference
Alexis Pinet, Bruno Figadère, Laurent Ferrié. Access to Functionalized 3,5-Disubstituted 1,2-Dioxolanes under Mild Conditions through Indium (III) Chloride Trimethylsilyl Chloride or Scandium (III) Triflate Catalysis. Advanced Synthesis & Catalysis 2020, volume 362, page 1190–1194.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


