Significance
Electrochemical routes for reducing CO₂ have been explored for a long time as a way to valorize a carbon source that is both abundant and chemically stubborn. The concept on paper is attractive: electrical energy goes in and reduced carbon products come out but experimentally it is not clean and despite clear gains in catalyst design, to translate those gains into reactor systems that function reliably beyond small, controlled demonstrations is challenging.The chemistry often looks promising, but the overall process falls short when viewed from an engineering standpoint and in many cases, the limitation is not catalytic activity but the surrounding environment, for example carbon dioxide must be supplied continuously at sufficient local concentration and in the same time reaction products must be removed before they interfere with further turnover. These requirements sound straightforward, but most commonly used reactor formats struggle to satisfy them at the same time. Batch cells are convenient for mechanistic studies, but they make inefficient use of the gas feed and mask transport limitations. Flow-based systems address some of these issues by increasing throughput, but they introduce others, particularly when alkaline electrolytes promote carbonate formation or destabilize long-term operation. Gas diffusion electrodes and membrane assemblies attempt to force closer contact between CO₂ and the catalyst surface. Under controlled conditions, they can work well. The problem is that they often rely on finely balanced parameters—wettability, pressure gradients, electrolyte composition—that are difficult to maintain over extended operation. Small deviations can lead to flooding, salt accumulation, or gradual loss of performance, which limits confidence in scale-up. Reactor design has largely followed catalyst development, rather than guiding it. Gas–liquid–solid contact is usually treated as something to be adjusted locally, not as the defining feature of the system. As a result, improvements in one metric tend to compromise another and until we address transport and reaction together at the reactor level, translating laboratory advances into practical CO₂ electroreduction technologies is not certain.
A recent research paper published in AIChE Journal and conducted by Dr. Qing Hu, Dr. Yujing Liu, Dr. Zhihang Wei, Dr. Linjie Chao, Dr. Lin Luo, and led by Professor Zhenmin Cheng from the School of Chemical Engineering at East China University of Science and Technology, the researchers developed a modular bubble-plate electrolyzer that embeds gas-facing electrocatalytic plates within a staged gas–liquid contactor. The system combines controlled bubble dispersion, repeated three-phase contact, and neutral aqueous electrolyte operation. Unlike conventional electrochemical cells, the reactor achieves high single-pass CO₂ conversion through structural staging rather than aggressive electrolyte or electrode design. The work establishes a scalable reactor framework adaptable to other gas-limited electrochemical reactions.
The research team designed a bubble-plate electrolyzer that integrates catalytic plates directly into a vertical gas–liquid contactor. The authors constructed the reactor as a modular column in which carbon paper electrodes coated with cobalt phthalocyanine intercept rising CO₂ bubbles which allowed the investigators to treat gas dispersion, liquid circulation, and electrochemical reaction as coupled processes rather than independent variables. The researchers demonstrated that gas redistribution at the base of the column broke large bubbles into finer populations, and suppressed channeling and promoting lateral mixing. As bubbles ascended, the authors observed sustained accumulation beneath each catalytic plate, which created repeated zones where gas, liquid electrolyte, and solid catalyst met and their computational flow analysis supported their findings and showed circulation patterns that continually refreshed these contact regions and limited stagnation. The study examined how catalyst placement influenced electrochemical behavior and compared gas-facing, liquid-facing, and double-sided catalyst configurations on otherwise identical plates. They observed that exposing the catalyst surface directly to the gas phase produced markedly higher CO selectivity and partial current than configurations dominated by liquid contact and when the authors isolated the gas-facing layer by sealing the opposite side of the electrode, performance remained high, indicating that the outer surface governed reaction rates more strongly than catalyst buried within the substrate. The researchers contrasted the bubble-plate electrolyzer with a conventional batch cell under matched conditions to separate geometric effects from transport effects and although increasing electrode area improved current in both systems, the authors showed that the bubble-plate configuration sustained substantially higher CO production at equivalent area. Time-resolved measurements revealed that continuous bubbling rapidly restored high selectivity after periods of CO₂ depletion, linking performance directly to gas delivery rather than intrinsic catalyst behavior.
The investigators then explored how reactor stacking altered conversion and by assembling multiple catalytic stages within the same column, the research team increased the effective reaction length without changing electrolyte composition or operating pressure. Additionally, the authors observed near-linear gains in single-pass CO₂ conversion as layers were added, while maintaining similar current–potential behavior at low to moderate current density. Experiments with mixed gas feeds allowed the researchers to distinguish between CO₂ depletion and product accumulation, leading them to conclude that local CO₂ concentration, not CO inhibition, governed observed trends. On top of that, the study examined gas flow rate as a control variable linking residence time and selectivity and demonstrated that slower gas flow increased conversion by extending contact time but reduced Faradaic efficiency when CO₂ became locally depleted. Faster flow preserved selectivity but shortened residence time. Indeed, by balancing these effects across staged plates, the researchers identified operating windows that combined high conversion with acceptable selectivity under neutral electrolyte conditions.
In conclusion, the work of Professor Zhenmin Cheng and co-workers demonstrated that the bubble-plate electrolyzer shows that controlling how gas interacts with catalytic surfaces can recover conversion efficiency under mild conditions. The ability to reach substantial single-pass conversion in dilute bicarbonate electrolyte directly addresses a long-standing barrier associated with carbonate formation and salt precipitation in alkaline systems. Moreover, the scientists showed that staged electrochemical reactors can decouple local reaction kinetics from overall conversion. By distributing reaction across multiple, well-mixed compartments, the system avoids the sharp concentration gradients that limit utilization in single-pass flow cells and brings electrochemical CO₂ reduction closer to established reactor models used in heterogeneous catalysis, where conversion is managed through residence time and contact efficiency rather than extreme operating conditions. We believe the implications of the innovation extend beyond CO production because the reactor concept provides a general framework for reactions limited by gas solubility or product removal, including other electrochemical transformations involving poorly soluble reactants. Because the architecture relies on modular plates and conventional materials, it provides a practical route for scaling without redesigning catalysts or electrolytes from first principles. Also, industrial relevance may depend less on pushing catalysts to theoretical limits and more on integrating them into reactors that respect transport constraints. The bubble-plate electrolyzer doesn’t eliminate trade-offs between conversion and selectivity, but it makes those trade-offs explicit and tunable through design variables familiar to chemical engineers. In doing so, it provides a credible bridge between laboratory electrochemistry and continuous chemical processing.
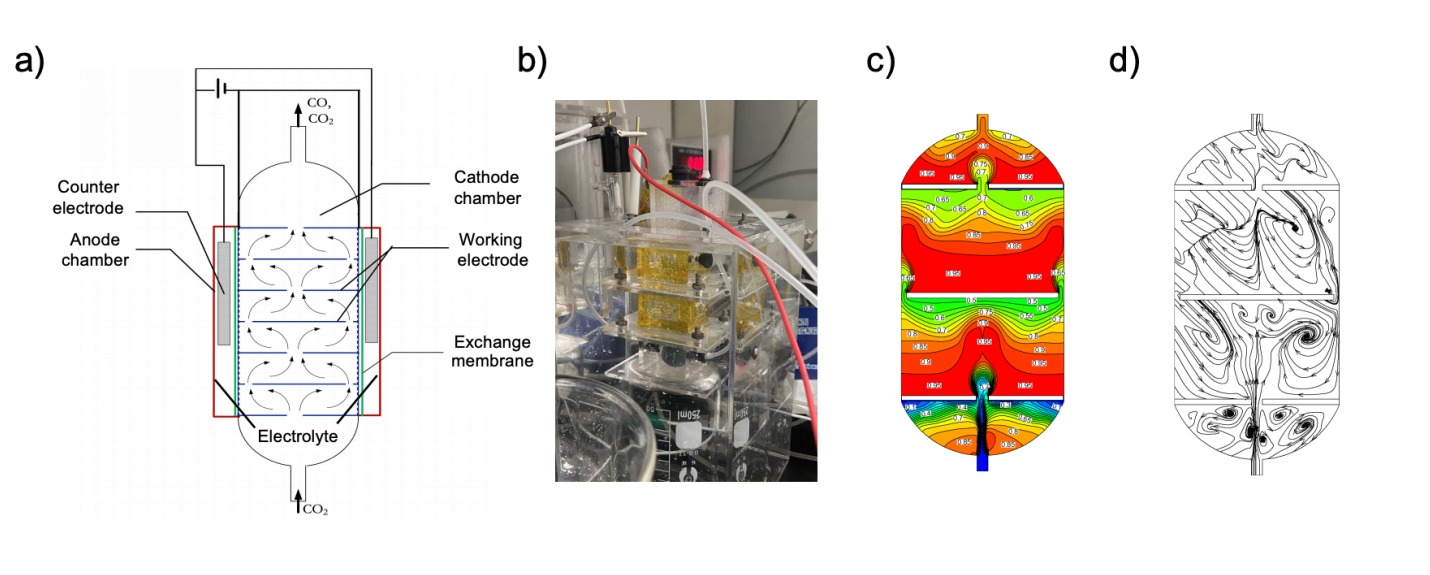
Figure 1 Design and flow visualization of the bubble-plate electrolyzer. (A) Schematic diagram of the electrolyzer. (B)Photograph of the experimental setup. (C) Two-phase flow volume fractions within the tower (label = liquid volume fraction). (D) The streamline of liquid within the tower. Copyright 2025 American Institute of Chemical Engineers
Reference
Hu, Qing & Liu, Yujing & Wei, Zhihang & Chao, Linjie & Luo, Lin & Cheng, Zhenmin. (2025). CO2 electroreduction in a bubble‐plate electrolyzer: A new route to scale up. AIChE Journal. 71. 10.1002/aic.18875.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

