Significance Statement
Two very simple operations in the macroscopic world are threading (for example of beads on a string) and weaving to make a two-dimensional structure. How can this be done at the atomic scale where the component molecules must assemble themselves without external manipulation? This paper reports the self-assembly of these two structures and shows the path followed to be quite different from the macroscopic world.
A threading reaction in the macroscopic world involves passing a long thread through a series of loops or beads. In the self-assembly approach, a short molecule (the stub) is threaded through one cyclic molecule (the loop) by coordination of loop and stub to a copper(I) ion. The stubs are then linked together by a second complexation reaction to form the thread (Fig. 1).
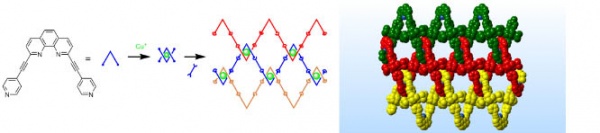
A woven structure is characterised by the crossings of different strands. In the macroscopic world this is achieved by twisting the strands around each other, but it would be very hard to do this with a series of long molecules. The self-assembly approach is not to start with the strands, and add the crossings, but to make the crossings and assemble them into strands. The crossings are made by binding two U-shaped molecules around them and then linking the crossings in a second reaction to give a structure reminiscent of a chain link fence (Fig. 2). An interesting observation is that each crossing is chiral (it cannot be superposed ion its own mirror image), and that the assembly takes place in such a way that every crossing in the structure has the same chirality.
Figure Legend 1. Schematic view of the construction of a necklace (left) with a view of the X-ray crystal structure on the right. Loops and thread are shown as red and green respectively.
Figure Legend 2. Construction of the interwoven chain link (left) with the X-ray crystal structure of the assembled structure (right). The different threads are distinguished as green, red and yellow.
Journal Reference
Chemistry – A European Journal, Volume 21, Issue 24, pages 8851–8858, June 8, 2015
Cristina Alvariño1,2,, Dr. Damien Simond1,, Pau Moneva Lorente1, Dr. Céline Besnard3 , Prof. Alan F. Williams1,
[expand title=”Show Affiliations”]- Department of Inorganic and Analytical Chemistry, University of Geneva, 30 quai Ernest Ansermet, CH 1211 Geneva 4 (Switzerland)
- Departamento de Química Fundamental, Universidade da Coruña, Facultad de Ciencias, Rúa da Fraga, 10. La Coruña (Spain)
- Laboratory for X-ray Crystallography, University of Geneva, 24 quai Ernest Ansermet, CH 1211 Geneva 4 (Switzerland).
Abstract
Assembly of two ditopic units, a phenanthroline substituted by 4-ethynyl pyridines at the 2-and 9-positions and a dimetallic paddlewheel, gives a linear chain polymer rather than a closed cyclic species, which would appear equally possible. The chain may be decorated by binding a copper-containing macrocycle around the phenanthroline units to form a polypseudorotaxane. When two phenanthroline ligands are assembled in a first step around copper(I), the paddlewheel acceptor can link them in a second step to form a two-dimensional interwoven grid that resembles the form of a chain-link fence. Each copper(I) centre in this structure is chiral, and the crystal shows complete homochirality, implying selection during the assembly process.
© 2015 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Go To Chemistry – A European Journal
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.