Significance Statement
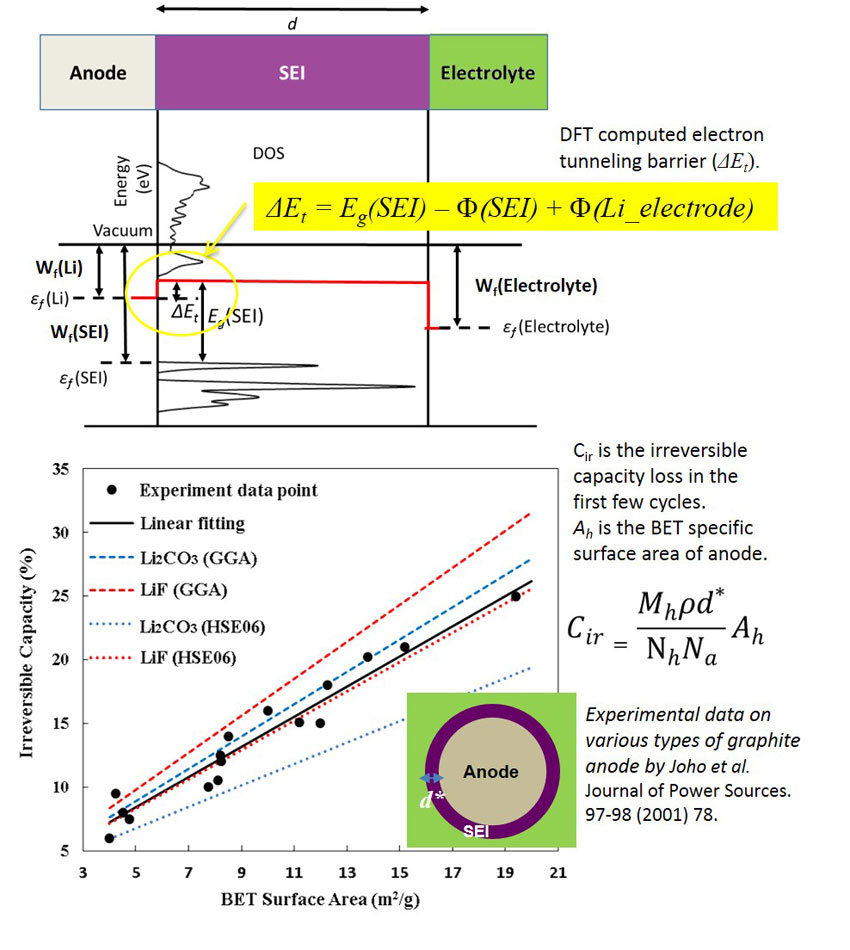
The life of lithium ion batteries is typically estimated by extrapolating accelerated life testing data. As a first step toward mechanism-based predictive battery life modeling, Lin et al. developed an analytical model, which takes electron tunneling properties obtained from quantum mechanical calculations as inputs, to estimate the first cycle irreversible capacity loss. (Y. Lin et al., J. Power Sources 309, 221-230, 2016) The predicted first cycle irreversible capacity loss shows good agreement with experimental results obtained on various carbon materials (Joho et al. Journal of Power Sources. 78, 97-98, 2001). This is a bit surprising, since no fitting parameters were used.
It is generally recognized that the formation and continuous growth of a solid electrolyte interphase (SEI) layers due to electrolyte decomposition are responsible for the irreversible capacity loss of batteries in both the initial and subsequent stages of cycling. In this study, a density function theory (DFT) – based electron tunneling model was used to assess the electronic insulating ability of inorganic solid electrolyte interphase components (Li2CO3, LiF, Li3PO4), such as electron tunneling barrier, critical thickness & initial irreversible capacity loss caused by Li+ ion consumption. These useful quantifiable parameters allow meaningful design principles for anode protection to circumvent cycling instability and capacity loss.
These calculations also show that the critical thickness needed to block electron tunneling on the electrode surface for inorganic solid electrolyte interphase components is ~2~3 nm. The coupling effect of stress, e.g. tension and compression induced by electrode volume expansion, on electron tunneling barriers was also investigated. It was predicted that electron tunneling barriers decrease under tension and increase under compression, causing non-negligible effect on the critical thickness & irreversible capacity loss due to deformation.
Since the initial irreversible capacity loss due to solid electrolyte interphase formation predicted by this fitting-parameter-free model agrees well with experiments, it suggests that the initial solid electrolyte interphase formation is likely to be controlled by the self-limiting electron tunneling property of the inorganic components in solid electrolyte interphase.
It also suggest that other electron transport mechanisms, such as polaron, defects, interfaces, cracked solid electrolyte interphase, and/or the instability of the outer solid electrolyte interphase components, such as Li2EDC and other radicals are still needed in order to explain the continuous growth of solid electrolyte interphase, which is responsible for the long term degradation of the batteries. Therefore, quantifying these mechanisms will eventually lead to a comprehensive life prediction model for lithium ion batteries.
This work is supported by the degradation science research theme as part of Nanostructures for Electrical Energy Storage (NEES), an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences under Award number DESC0001160 and NSF GOALI under CMMI-1235092.
Journal Reference
Yu-Xiao Lin1, Zhe Liu2, Kevin Leung3, Long-Qing Chen2, Peng Lu4, Yue Qi1 . Connecting the irreversible capacity loss in Li-ion batteries with the electronic insulating properties of solid electrolyte interphase components. Journal of Power Sources,Volume 309, 2016, Pages 221–230.
[expand title=”Show Affiliations”]- Department of Chemical Engineering and Materials Science, Michigan State University, East Lansing, MI 48824, USA
- Department of Materials Science and Engineering, Penn State University, University Park, PA 16802, USA
- Sandia National Laboratories, Albuquerque, NM, USA
- General Motors Research and Development Center, Warren, MI 48090, USA
Go To Journal of Power Sources
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



