Significance
Following recent technological advances, research on the effects of loading zeolites with ion-exchanged or impregnated ions or clusters of two metals has shown that their catalytic activity, stability and selectivity becomes greatly affected. In addition, recent publications have reported on the different scenarios of the two metals’ distribution, chemical state and potential competitive siting. Such studies have pointed out that metal ions with higher charge density occupy small cages and normally prevent another ion migration from supercages. Moreover, it has been seen that preferential ion locations depend on the type of zeolite used. Recently, only T3 sites located in 8-membered ring channels have been observed to selectively perform dimethyl ether carbonylation to methyl acetate, as the others catalyze side reactions and lead to a fast deactivation. Therefore, it is important that the synergetic effects of zinc and copper ions be comprehended since the reaction is of much significance as it offers an iodine-free route to acetic acid or ethanol from dimethyl ether that could be obtained either from methanol or directly from a syngas.
To this note, University of Alberta researchers Allen Reule, Dr. Jing Shen and Professor Natalia Semagina from the Department of Chemical and Materials Engineering assessed the locations of copper and zinc species in the bimetallic ion exchanged mordenite. They hoped the results would potentially explain the previously observed catalytic behavior in dimethyl ether carbonylation that would in turn contribute to the understanding of bimetallic ion exchange on a zeolite. Their work is currently published in the research journal, Chem.Phys.Chem.
In brief, the research method used entailed the characterization of samples comprised of various copper and zinc ratios, by diffuse-reflectance infrared Fourier-transform spectroscopy. Next, pore distribution analysis through argon physisorption, X-ray photoelectron spectroscopy, temperature-programmed reduction and transmission electron microscopy were carried out for the test samples.
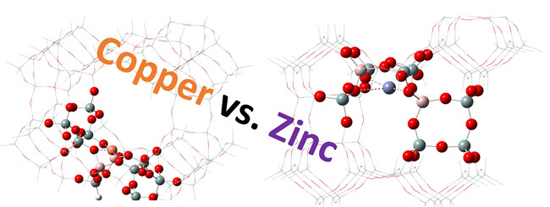
The authors observed that in the bimetallic samples, zinc addition resulted in an increase in the coordination strength of copper to the zeolite and prevented copper (II) oxide nanoparticle formation without copper-zinc alloying. Additionally, they noted that copper preferentially exchanged into mordenite as compared to zinc thereby affecting the favorable location of zinc ion-exchange. All in all, the researchers recorded that the effect was achieved at 0.25 copper-zinc molar ratio for the mordenite with a specific silicon-aluminum ratio.
In a nutshell, Professor Natalia Semagina and her colleagues presented the analysis of mordenite samples ion exchanged with copper and zinc ions for the purposes of comprehending metal distribution and its effect on dimethyl ether carbonylation. Generally, it was seen that zinc alone readily exchanged into 8-MR structures (T3 sites), but in the presence of copper, it preferred to exchange into T4 sites in 12-MR structures as opposed to the T3 atoms. Altogether, this led to the conclusion that the selective blockage of T4 sites inhibited the formation of the trimethyl oxonium ion from the adsorbed methyl group and dimethyl ether, and increased the catalyst lifetime during dimethyl ether carbonylation.
Reference
Allen A. C. Reule, Jing Shen, Natalia Semagina. Copper Affects the Location of Zinc in Bimetallic Ion-Exchanged Mordenite. Chem.Phys.Chem 2018, volume 19, page 1500– 1506.
Go To Chem.Phys.Chem Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

