Significance
Carbon monoxide (CO) is considered as a highly toxic gas to humans and animals. It rapidly binds with the hemoglobin thus decreasing the oxygen transportation capacity in the blood resulting in tissue damage. However, carbon monoxide which is a waste resulting from steel and other metallurgical plants is a useful raw material for the manufacturing of numerous chemicals such as methanol. It is found in a mixture of gases produced by these plants. The gases include nitrogen, methane, hydrogen and carbon dioxide. Therefore, an efficient method for the separation of CO from the gas mixture has attracted significant attention among researchers and industrialists.
Among the available separation techniques, adsorption, cryogenic separation and absorption are widely used. However, they have drawbacks that affect their efficiency. For instance, cryogenic separation is deemed unfit when the gas mixture contains nitrogen due to the small difference in the boiling points while absorption leads to corrosion of the equipment leading to additional cost. However, adsorption techniques have been qualified as the best especially when a suitable adsorbent material can be developed. Since the gases’ mixture contains CO2 which has a higher polarization ability as compared to CO, the adsorbent material in question should be of high stability, large CO selectivity and working capacity.
Recently, a group of researchers at Yonsei University: Ah-Reum Kim, Tae-Ung Yoon, Seung-lk Kim and Professor Youn-Sang Bae synthesized a highly selective CO adsorbent for its efficient separation from a mixture of several gases. Their main aim was to enhance CO/CO2 selectivity as well as CO working capacity in the adsorbent material. Their work is published in the research journal, Chemical Engineering Journal.
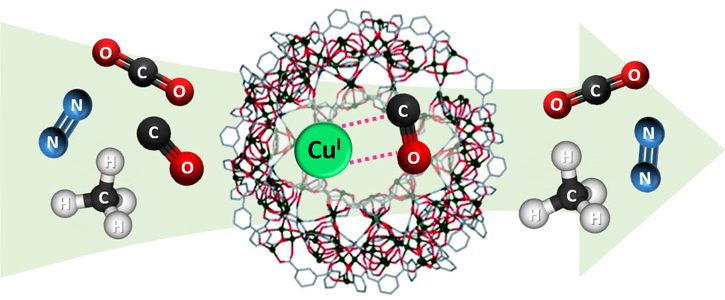
Briefly, the authors commenced their experimental work by loading Cu(II) species into the pores of an iron-based redox-active metal-organic framework. Consequently, the Fe(II) sites in the pores were used under mild conditions to reduce the Cu(II) species to Cu(I) species without the need of high-temperature calcination or a reducing agent. Furthermore, samples of Cu(I)-loaded MIL-100(Fe) with different copper contents were synthesized by impregnating CuCl2 and reducing them to CuCl. Eventually, various methods were used to analyze the loading of Cu(I) species and the role of Fe(II) sites.
The authors observed that the resulting adsorbent material exhibited significantly improved CO working capacity and consequently CO/CO2 selectivity as compared to the pristine adsorbent materials. In addition, 0.9Cu@MIL-100 exhibited high CO/CH4 and CO/N2 selectivities.
The study is the first to present an adsorbent material with remarkably high CO/CO2 selectivity and CO working capacity. Therefore, it can be used for the separation of carbon monoxide from various gas mixtures. For instance, high-purity CO can be obtained from a mixture including CO, CO2, CH4, N2 and H2 under dynamic mixture flow conditions. Also, it can be regenerated under mild conditions. Furthermore, the study provides insights towards the synthesizing of even better and suitable adsorbents for industrial use as it is also environmentally friendly.
Reference
Kim, A., Yoon, T., Kim, S., Cho, K., Han, S., & Bae, Y. (2018). Creating high CO/CO2 selectivity and large CO working capacity through facile loading of Cu(I) species into an iron-based mesoporous metal-organic framework. Chemical Engineering Journal, 348, 135-142.
Go To Chemical Engineering Journal Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.





