Significance
Stimuli-responsive porous materials have diverse and innovative applications across various fields due to their unique ability to change properties or behavior in response to external stimuli. These materials can undergo reversible transformations in response to external stimuli such as light, heat, guest molecules, magnetism, force, and pH. This ability to switch between different configurations or states has found applications in chemical sensors, mechanical actuators, anti-fake inks, and smart adsorbents, among others. For instance, these materials can be used in targeted drug delivery systems. They respond to specific stimuli such as pH, temperature, or biochemical signals to release drugs at targeted sites in the body, enhancing treatment efficacy and reducing side effects. Their ability to respond to environmental changes makes them ideal for sensors and biosensors. They can detect changes in pH, temperature, humidity, or the presence of specific molecules, and are used in medical diagnostics, environmental monitoring, and industrial process control. In biomedical engineering, stimuli-responsive porous materials can be used to create scaffolds that adapt to biological environments, promoting cell growth and tissue regeneration. They can respond to biological signals to aid in healing or regeneration processes. These materials can be used in the capture and removal of pollutants from air and water. Their porous nature allows for efficient adsorption, and their stimuli-responsive properties can be used to release and recover the pollutants in a controlled manner for easier disposal. Stimuli-responsive porous materials can also used to create smart coatings and surfaces that can change properties like color, permeability, or texture in response to environmental changes, which is useful in applications ranging from smart windows to protective coatings. They also can find applications in batteries and fuel cells where they can respond to electrical, thermal, or chemical stimuli to improve efficiency and durability of energy storage and conversion systems. Moreover, they can be used as catalysts or catalyst supports. Their ability to change properties like surface area or chemical functionality in response to external stimuli can be used to enhance catalytic reactions or control reaction pathways. These applications highlight the versatility of stimuli-responsive porous materials in addressing challenges across various sectors, from healthcare to environmental sustainability. The majority of stimuli-responsive porous materials developed to date have relied on mechanisms involving molecule isomerism or host-guest interactions. However, a new study published in the Angewandte Chemie International Edition journal by graduate student Ze-Jiu Diao, Si-Yi Liu, Hui Wen, Dr. Guoliang Liu, Tao Yang, Jing-Jing Li, Prof. Xiao-Qin Liu, and led by Professor Lin-Bing Sun developed a novel approach to stimuli-responsive porous materials based on a detachment mechanism.
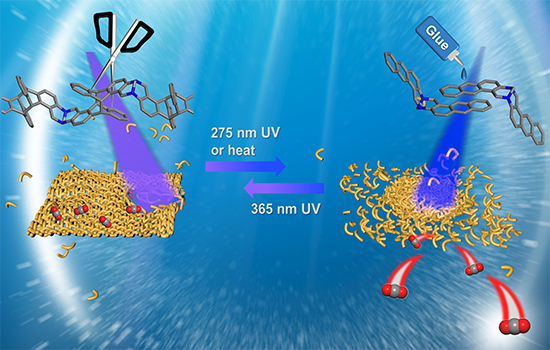
Detachable Porous Polymer (DT-POP-1) was constructed from an anthracene-containing monomer (AnMon) via a reversible [4π+4π] cycloaddition reaction initiated by irradiation with 365 nm UV light. What makes DT-POP-1 unique is its ability to detach into the monomer AnMon when exposed to 275 nm UV light or heat. Furthermore, it can be re-polymerized into DT-POP-1 by re-irradiation with 365 nm UV light. This polymerization/detachment process is entirely reversible. Due to the irregular packing of polymer chains with a contorted structure, DT-POP-1 boasts substantial porosity, with a high Brunauer-Emmett-Teller (BET) surface area of 554 m²/g, significantly higher than that of the monomer AnMon after detachment, which only has a surface area of 10 m²/g. This difference in porosity is pivotal to the material’s effectiveness in adsorbing gases, particularly carbon dioxide (CO2).
The authors synthesized monomer AnMon through Tröger’s base condensation reaction, and its structure was confirmed through various spectroscopic techniques, including 1H NMR, 13C NMR, and Fourier transform infrared spectra. DT-POP-1, on the other hand, was synthesized from AnMon under 365 nm UV light irradiation and was characterized through techniques such as 1H NMR, UV/Vis absorbance spectra, gel permeation chromatography, differential scanning calorimetry, and thermogravimetric analysis. DT-POP-1’s solubility in organic solvents facilitated the determination of its molecular weight, which was found to be 6239 g/mol with a polydispersity index of 1.63.
The authors showed that DT-POP-1’s detachment can be induced not only by light irradiation but also by heat. The photodetachment process, initiated by 275 nm UV light, was monitored through 1H NMR and UV/Vis absorbance spectra, confirming the transformation of DT-POP-1 into AnMon. Moreover, the thermal detachment of DT-POP-1, which occurs at 449 K, was demonstrated through various analytical techniques, including 1H NMR, in situ diffuse reflectance infrared Fourier transform spectrum, and thermal analysis. The reversible polymerization/detachment cycle was confirmed, indicating that this process can be controlled by both light and heat stimuli.
One of the most significant aspects of DT-POP-1’s stimuli-responsive behavior is its gas adsorption performance, particularly in capturing CO2. The material’s rich microporosity, coupled with the presence of nitrogen atoms, makes it a promising candidate for CO2 capture. Adsorption isotherms showed that DT-POP-1 exhibited a remarkable CO2 adsorption capacity of 41.6 cm³/g at 273 K and 1 bar, with minimal N2 adsorption, resulting in a high CO2/N2 selectivity of 60 based on ideal adsorbed solution theory.
Furthermore, the researchers monitored the changes in CO2 adsorption capacity during the detachment process. After irradiation with 275 nm UV light, the adsorption capacity gradually decreased, consistent with the detachment process. Even after detachment was complete, DT-POP-1 retained some ability to adsorb CO2 due to partial retention of the stacking structure. Similarly, the material’s CO2 adsorption capacity decreased significantly when subjected to thermal detachment at 453 K, demonstrating the reversibility of its adsorption behavior. Cycle experiments further confirmed the material’s ability to maintain its adsorption capacity over multiple polymerization/detachment cycles.
In conclusion, the study by Professor Lin-Bing Sun and colleagues presents an important advancement in stimuli-responsive porous materials. DT-POP-1, constructed through a detachment mechanism, offers a unique combination of stimuli-responsiveness, high porosity, and reversible adsorption behavior. This material’s ability to switch between porous and non-porous states in response to external stimuli opens up new possibilities for various applications, including adsorptive separation, guest inclusion, and drug delivery. The innovative approach presented in this study represents a significant step forward in the development of stimuli-responsive materials and holds promise for addressing environmental and technological challenges in the future.
Reference
Diao ZJ, Liu SY, Wen H, Liu G, Yang T, Li JJ, Liu XQ, Sun LB. Detachable Porous Organic Polymers Responsive to Light and Heat. Angew Chem Int Ed. 2023 ;62(21): e202301739. doi: 10.1002/anie.202301739.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




