Significance
Hydrogen gas is a renewable and ecofriendly source of energy which releases water as its final product. Specifically, it has the highest gravimetric calorific value of all possible fuels. Hydrogen is also light and can be liquified in pressurized tanks hence increased storage capacity. Regardless, it has a critical drawback that severely limits it commercial use: that is, its low volumetric density under ambient conditions. Possible solutions to this shortcoming have been put forward over the years with physisorption of the gas in a dense adsorbed layer on materials of high surface area for efficient and safe storage systems being the most auspicious. As a result, the development of novel porous materials possessing extremely high surface areas and tunable pore sizes, such as metal–organic frameworks, has been the focus of much research overtime. Unfortunately, much of these researchers have concentrated on optimizing the hydrogen storage performance of metal–organic frameworks by increasing the specific surface area, which ultimately enlarges the gravimetric capacity at the expense of the volumetric capacity.
To this note, Rafael Balderas-Xicohténcatl (PhD candidate) and Dr. Michael Hirscher (group leader) at Max Planck Institute for Intelligent Systems in collaboration with Dr. Phillip Schmieder, Dr. Dmytro Denysenko, and Dr. Dirk Volkmer at Augsburg University compared directly the gravimetric and volumetric hydrogen storage capacity using one non-interpenetrated and one interpenetrated metal-organic framework. In their study, they successfully showed an improvement of the volumetric capacity by interpenetrating the framework structures. Their work is currently published in the research journal, Energy Technology, and highlighted as front cover in the March 2018 issue.
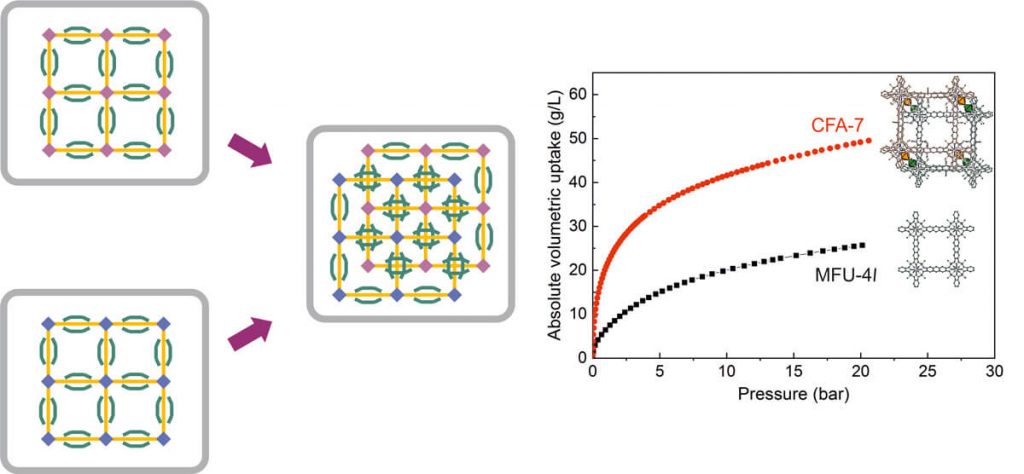
The research method employed involved the use of an automated Sieverts type apparatus with a micro-doser designed to measure the storage properties accurately with only small amount of sample (about 100 mg). Next, they performed the pressure-composition-temperature measurements with hydrogen gas up to a maximum pressure of 20 bar. Lastly, they experimentally assessed the influence of interpenetration on the volumetric and gravimetric hydrogen storage capacity by comparing two metal-organic frameworks of the Metal-Organic Framework Ulm University (MFU-4) family, non-interpenetrated MFU-4l and Coordination Framework Augsburg University-7 (CFA-7) with an interpenetrated structure.
The authors observed that from the comparison undertaken, CFA-7 showed over twice the volumetric storage capacity of MFU-4l. Additionally, the average heats of adsorption for MFU-4l and CFA-7 were determined to be 3.92 and 5.2 kilo joules per mole, respectively. Furthermore, they found out that the volumetric surface area, i.e. surface area per unit volume, available for the physisorption of gases was soley responsible in governing the volumetric storage capacity of porous physisorption materials.
In summary, Dr. Michael Hirscher and colleagues study presented a direct comparison of gravimetric and volumetric hydrogen storage capacity. Their main observation was that at 77 K the absolute volumetric hydrogen uptake of CFA-7 was more than twice that of MFU-4l. Altogether, their results illustrate clearly that the volumetric hydrogen storage capacity in porous materials can be drastically improved by using interpenetrated metal-organic frameworks.
Reference
Rafael Balderas-Xicohténcatl, Phillip Schmieder, Dmytro Denysenko, Dirk Volkmer, Michael Hirscher. High Volumetric Hydrogen Storage Capacity using Interpenetrated Metal–Organic Frameworks. Energy Technology 2018, volume 6, page 510 – 512.
Balderas‐Xicohténcatl Dr. Maurice Schlichtenmayer Dr. Michael Hirscher. Volumetric Hydrogen Storage Capacity in Metal–Organic Frameworks. Energy Technology 2018, volume 6, page 578 – 582.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



