Significance
Recent global trends towards a fossil-fuel-free society has yielded the rapid soar in demand of electrically powered systems. Specifically, the demand for battery powered systems has fueled the desire to have better performing batteries. In this view, lithium sulfur batteries are at the forefront of this revolution owing to their high theoretical specific capacity, high energy density, cost viability, and non-toxic characteristics. Unfortunately, practical application of this Li-S batteries remains a challenge due to issues such as the low conductivity of elemental sulfur and its discharge products, large volumetric changes during each electrochemical cycle, and the dissolution of polysulfide intermediates, which is termed the “shuttle phenomenon”.
Various approaches have been put forth as walk around of these drawbacks. Each approach has so far enhanced performance, selectively improved the electrical/ionic conductivity, inhibited the structural breakdown of the electrode or retarded the dissolution of polysulfide. Nonetheless, no countermeasures have been able to effectively contain the inevitable release of polysulfide via dissolution, an effect that seriously compromises the performance of Li-S cells.
In a recent publication, Prof. Woo Young Yoon together with Dr. Byung Dae Son, Sung Ho Cho, Ki Yoon Bae and Byung Hyuk Kim, from the Department of Materials Science and Engineering, at Korea University focused on fabricating an interlayer with ferroelectricity via a facile technique in a bid to resolves the outstanding standoff. This technique involved the construction of a ferroelectric nanoparticle-entangled carbon nanotube structure. Their work is currently published in the research journal, Journal of Power Sources.
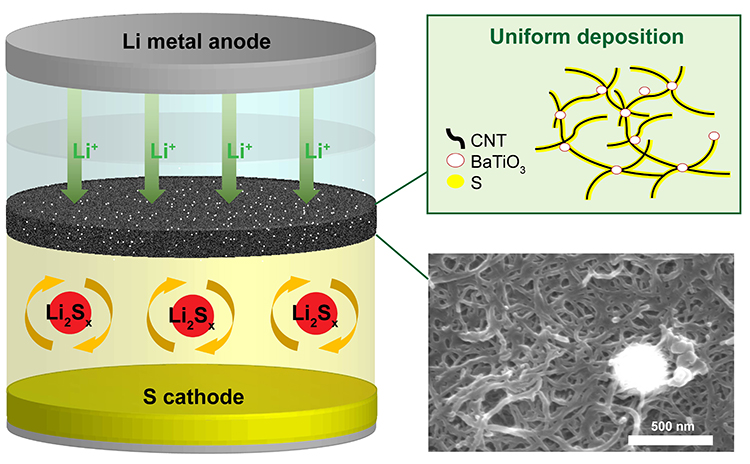
In brief, a carbon nanotube sheet having homogeneously distributed ferroelectric nanoparticles (BaTiO3) was applied to the Li-S cell system as a pseudo current collector between the cathode and separator. The prepared system was then subjected to electrochemical characterization among other tests.
The authors observed that the cell utilizing the ferroelectricity embedded interlayer exhibited a higher capacity at 0.2C than that of carbon alone at 200th cycle. Furthermore, it was confirmed that the retention of the coulombic efficiency was effectively maintained in long cycles at 0.5C. By analyzing the voltage profile and cyclic voltammetry results, it is observed that less polarization with effective charge transferring has occurred when ferroelectricity is applied to the current collector system. And through surface analysis on the interlayers with SEM and XPS after serial reactions of Li-S, it is confirmed that the uniform dispersion of active material with enhanced adsorption energy on the carbon matrix was enabled by ferroelectricity modification which contributed to the improvement in the electrochemical property during the charge/discharge reactions. Also, the decreased amount of polysulfide elution was detected after the modification.
In summary, an interlayer modified with a uniform distribution of BaTiO3 was introduced as a pseudo current collector in a Li-S cell, to capture the polysulfide intermediates that dissolve from the sulfur cathode and allow the eluted active material to continue the electrochemical reaction. Generally, it was reported that the effect of the ferroelectricity in the interlayer was not only holding the polysulfide as reported before, but also helping carbon current collector to distribute the uniform sulfur with increased binding energy between the final product and the current collector. Altogether, the evenly distributed polarization leads to a uniform deposition of sulfur, which results in the prevention of inactive sulfur agglomeration and dissolution of polysulfide, thereby pointing out that the utilization of active material can be improved with stabilized reaction.
Reference
Byung Dae Son, Sung Ho Cho, Ki Yoon Bae, Byung Hyuk Kim, Woo Young Yoon. Dual functional effect of the ferroelectricity embedded interlayer in lithium sulfur battery. Journal of Power Sources, volume 419 (2019) page 35–41.
Go To Journal of Power Sources Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

