Significance
Gold catalysis is a cornerstone in contemporary organic synthesis, known for its unparalleled reactivity and selectivity in activating alkynes. However, the activation of gold catalysts, particularly the Au−Cl bond in [AuCl(L)] complexes, has long been a challenge, often requiring the use of silver salts or other activators. To simplify the catalyst activation process and expand the utility of gold catalysis in organic synthesis, with the aim of providing a sustainable and efficient pathway for the synthesis of complex organic compounds, PhD candidate Nikolaos Tzouras and Professor Steven P.Nolan from Ghent University in collaboration with Leandros Zorba, Entzy Kaplanai, Nikolaos Tsoureas and Professor Georgios Vougioukalakis from the National and Kapodistrian University of Athens and Dr. David Nelson from the University of Strathclyde published detailed investigation in ACS Catalysis into the use of hßexafluoroisopropanol (HFIP) as a novel agent in gold-catalyzed cycloisomerization reactions and sequential transformations, focusing on its dual role as both a solvent and an activator, which is a significant departure from traditional methodologies that often require external activators or additives.
HFIP is a fluorinated alcohol known for its unique properties, such as high polarity, ability to stabilize cations, and its role as a strong hydrogen bond donor. These characteristics make HFIP an excellent solvent and reagent in various chemical reactions and processes. The central premise of their research was to explore the capability of HFIP to activate gold catalysts, specifically the Au−Cl bond in gold(I) chloride complexes, through hydrogen bonding. This approach aimed to bypass the conventional need for silver salts or other additives, which can complicate the reaction process and affect the purity of the final product.
Through comprehensive detailed experimentation, the team screened various ancillary ligands and counterions to optimize the conditions for gold-catalyzed cycloisomerizations. This involved evaluating the effectiveness of different [AuCl(L)] complexes in the presence of HFIP, determining optimal catalyst loadings, and fine-tuning reaction parameters to achieve high yields and selectivity. Additionally, they applied the HFIP-assisted gold catalysis to a range of cycloisomerization reactions, including the transformation of propargylamides, ynoic acids, and aryl propargyl ethers. These reactions were chosen for their relevance in organic synthesis and their ability to produce a variety of valuable organic compounds, such as 2H-chromenes, functionalized ketoesters, and compounds with bioactive indole cores.
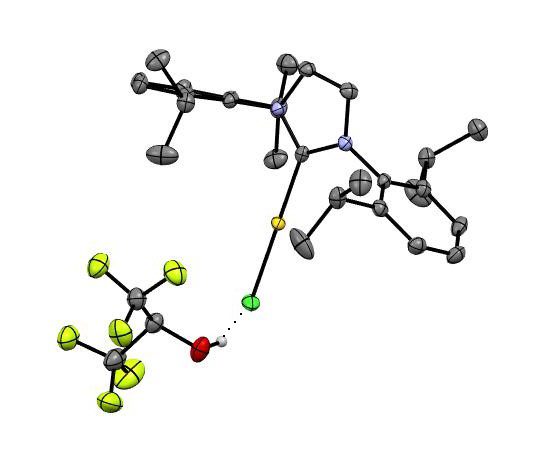
To understand the underlying mechanism, the researchers conducted in-depth mechanistic studies, including capturing a “snapshot” of the crucial hydrogen bonding interaction between HFIP and the gold catalyst in the solid state. This was complemented by density functional theory (DFT) calculations, which provided further insights into the interaction dynamics and the activation process. One of the most significant findings was that HFIP could effectively activate gold catalysts without the need for external activators or additives. This simplifies the catalytic system, making the reactions more straightforward and potentially more environmentally friendly. The authors demonstrated that HFIP-assisted gold catalysis could achieve high catalytic efficiency with minimal catalyst loading, in some cases down to parts per million levels. This represents a substantial improvement over traditional gold-catalyzed reactions, which often require higher concentrations of catalysts. The methodology proved to be versatile and broadly applicable to various cycloisomerization reactions. This opens up new possibilities for the synthesis of complex organic molecules, which are valuable in diverse fields such as pharmaceuticals, agrochemicals, and materials science. The detailed mechanistic studies provided valuable insights into how HFIP activates the gold catalyst through hydrogen bonding. This contributes to a deeper understanding of gold catalysis and could inform the design of new catalytic systems and reaction pathways.
Overall, the study by Nikolaos Tzouras, Steven Nolan, and their collaborators marks a significant advancement in the field of gold catalysis, introducing HFIP as a powerful tool for the activation of gold complexes and the facilitation of cycloisomerization reactions. The findings not only enhance the understanding of gold catalysis but also offer a practical and efficient approach to organic synthesis, aligning with the goals of green chemistry and sustainability.
Reference
Nikolaos V. Tzouras, Leandros P. Zorba, Entzy Kaplanai, Nikolaos Tsoureas, David J. Nelson, Steven P. Nolan*, and Georgios C. Vougioukalakis*. Hexafluoroisopropanol (HFIP) as a Multifunctional Agent in Gold-Catalyzed Cycloisomerizations and Sequential Transformations. ACS Catal. 2023, 13, 13, 8845–886
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


