Significance Statement
The current study is motivated by the challenges of finding renewable polymers and polar monomers that can be polymerized with coordination initiators. Currently, polyethylene as well as it copolymers are produced mainly from petroleum as well as natural gas. The two sources are considered unsustainable raw materials. Fortunately, renewable polyethylene can be produced from sugarcane as a raw material, and this procedure has already been commercialized.
In the case of ethylene, copolymers and comonomer should be renewable specifically if its content is high. Ethylene copolymers with high amounts of polar comonomers have potential as coatings, laminates, and adhesives. The produced polymer composed of polar groups can be post-modified, which means that the polar groups can be converted to other functional groups or implemented for crosslinking or grafting.
Many synthetic copolymers can be produced through radical polymerization that demands higher temperature as well as pressure as compared to coordination initiated polymerization. Single-site catalysts are beneficial in offering improved control over the polymerization conditions and polymer attributes, for instance better control over molecular weight distribution. Unfortunately, the polymerization of polar monomers by coordination catalysts comes with a challenge as a number of transition metals form stable bonds to electronegative atoms including nitrogen and oxygen.
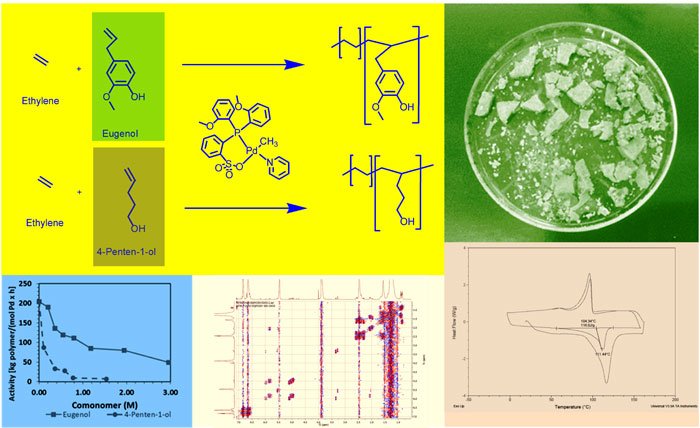
Late transition metal catalysts have been found to be more tolerant to oxygen containing monomers. Palladium aryl sulfonate catalysts, which have been incepted more recently, can be implemented to polymerize numerous types of highly polar monomers with appreciable comonomer incorporation. Researchers led by Professor Massoud Miri at The Rochester Institute of Technology performed a number of binary copolymerization of ethylene with two comonomers with palladium aryl phosphine sulfonate catalysts. The two comonomers were eugenol and 4-penten-1-ol. Their research work is published in Polymer.
The proposed ethylene copolymerizations indicated that the renewable comonomer eugenol was superior in several aspects over 4-penten-1-ol as comonomer. Eugenol polymerized at a significantly higher activity and led to less chain transfer resulting in slightly higher molecular weights. It was observed to be incorporated more easily into the polymer and within the polymer chains rather than as end groups. Irrespective that both monomers had a similar distance between the hydroxyl group and the terminal alkene group, the aromatic group acted as more effective spacer group.
Eugenol was inserted into copolymer chains through 2,1 as well as 1,2 additions, while 4-penten-1-ol was polymerized mostly by 2,1 additions and formed a considerable portion of the end groups. In addition, the authors observed that the eugenol copolymers had antibacterial attributes in contrast to polyethylene or the ethylene/4-penten-1-ol copolymers. The ethylene/eugenol copolymers therefore would be particularly useful for food or medical packaging.
The study presents a successful production of copolymers of ethylene with eugenol implementing a single component palladium aryl sulfonate catalyst.
Reference
L.R. Parisi, D.M. Scheibel, S. Lin, E.M. Bennett, J.M. Lodge, and M.J. Miri. Eugenol as renewable comonomer compared to 4-penten-1-ol in ethylene copolymerization using a palladium aryl sulfonate catalyst. Polymer, volume 114 (2017), pages 319-328.
Go To Polymer Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


