Significance
An Impressive and Novel Process for Liquefaction of Lignocellulosic Biomass
Considering the environmental impact of fossil resources, the exploration of alternative renewable energy sources has rapidly increased. Lignocellulosic biomass, in particular, has exhibited the great potential of producing biochemical and value-added biofuels. Levulinic acid is an example of the platform chemicals produced through liquefaction of biomass. Nevertheless, decomposition of cellulose or hexose into levulinic acid requires catalysts such as mineral acids, resulting in additional challenges such as undesirable side reactions, difficulty in separation, and high equipment corrosion. Alternatively, heterogenous catalysts developed for effective conversion of carbohydrates into levulinic acid in water faces numerous challenges, including low catalytic activities and complex preparation processes that hinder their industrial applications. Thus, the development of economic and sustainable catalytic systems is highly desirable.
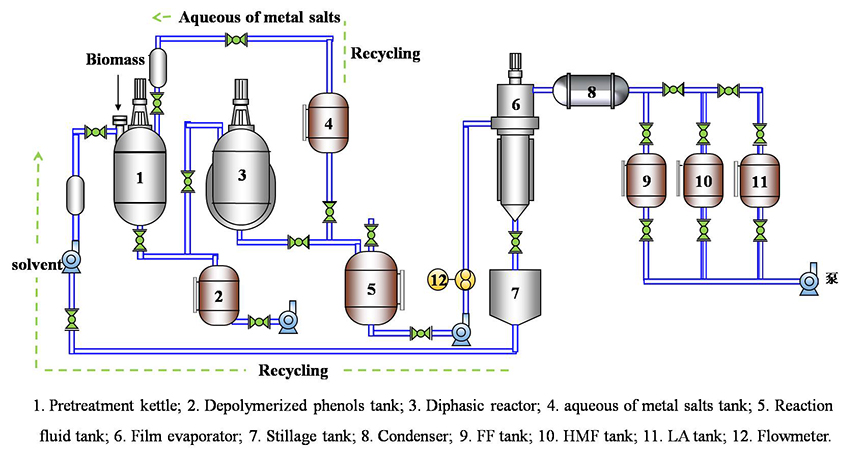
Production of levulinic acid from biomass using low-cost metals has recently attracted considerable attention. Researchers at the Institute of Chemical Industry of Forest Products, Chinese Academy of Forestry: Dr. Chao Liu, Dr. Min Wei, Dr. Jia Wang, Prof. Junming Xu, Prof. Jianchun Jiang, and Prof. Kui Wang developed a facile and effective process for converting cellulose and waste lignocellulosic residues into levulinic acid in a polar aprotic solvent using low-cost sulfate catalysts. In their research, they investigated how the synergistic effects work between the solvent and catalysts. Their work is currently published in the research journal, ACS Sustainable Chemistry & Engineering.
The research team obtained more than 77% mol and 61% mol of biochemicals from the cellulose and bamboo meal wastes, respectively, with a corresponding 95.4 wt% cellulose conversion. The reaction time and temperature exhibited considerable effects on the cellulose conversion and levulinic acid yield. At a constant temperature of 200°C, levulinic acid yield increased gradually alongside reaction time that ranged between 1-2 hours. Temperatures above 200°C resulted in low conversion and yield. NMR spectroscopy analysis revealed significant cellulose depolymerization under the cationic polarization of polar aprotic solvents. Moreover, the destruction of cellulose and restraint of humin formation was attributed to the existence of water.
Notably, all the solvents and byproducts obtained during the process could be efficiently separated and collected via the distillation process. Besides, these solvents and catalysts could be recycled without a significant reduction in the conversion rate or chemical yields, thus reducing the overall process costs. Finally, the authors validated the feasibility of the process through a real-life lignocellulosic application. The outstanding performance of the liquefied system was a good indication of its potential industrial applications.
In summary, the study demonstrated an impressive and novel liquefied process for value-added utilization of lignocellulosic biomass with attractive industrial application prospect. The outstanding performance of the process was attributed to the synergistic effects between the active acid and cation polarization. Besides, the byproducts could be easily separated from the solvents and target products and recycled without a significant reduction in the yield or conversion rates. Altogether, Professor Kui Wang and his colleagues mentioned their study would open ways for detailed investigation of the synergistic effects of different intermediates formed during liquefaction of lignocellulosic biomass for efficient industrial applications.
Reference
Liu, C., Wei, M., Wang, J., Xu, J., Jiang, J., & Wang, K. (2020). Facile Directional Conversion of Cellulose and Bamboo Meal Wastes over Low-Cost Sulfate and Polar Aprotic Solvent. ACS Sustainable Chemistry & Engineering, 8(14), 5776-5786.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


