Significance
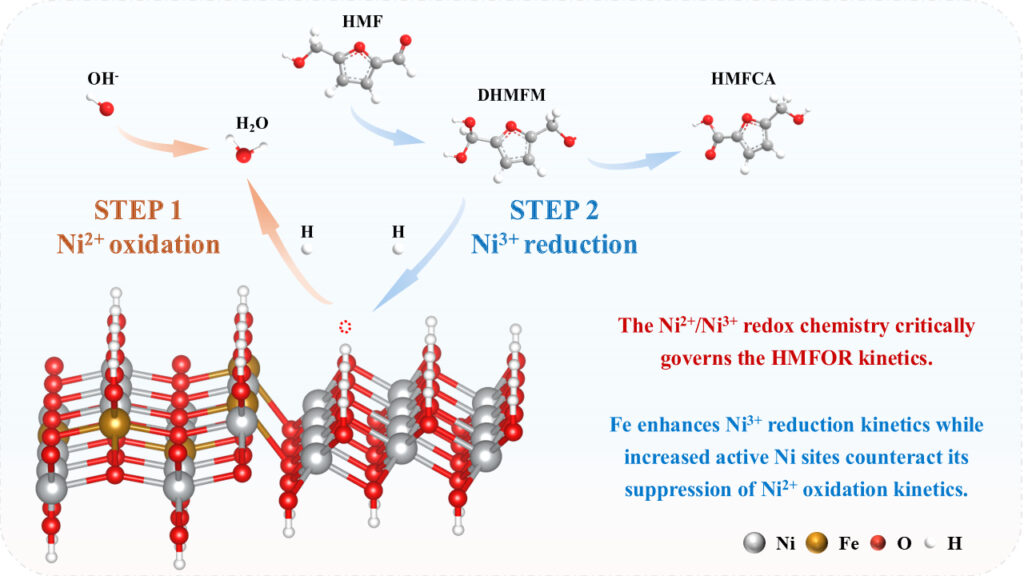
Electrochemical oxidation of biomass-derived molecules is an important alternative to thermochemical routes because it promises chemical selectivity under conditions that don’t rely on elevated temperature, pressure, or external oxidants. For 5-hydroxymethylfurfural: it is chemically dense, has both aldehyde and hydroxymethyl groups on a furan ring, and each functionality opens parallel oxidation sequences that compete rather than cooperate and controlling those sequences without sacrificing efficiency has proven difficult, even when the thermodynamic driving force is sufficient. Nickel-based hydroxides and oxyhydroxides are appealing because of their surface adsorption and from a redox cycle in which Ni(II) and Ni(III) interconvert under anodic bias. That redox chemistry enables hydrogen abstraction steps that are otherwise sluggish in alkaline media. The field has long treated Ni(II) oxidation as the dominant bottleneck, and a large fraction of prior design logic follows directly from that assumption. Dopants, defect engineering, and surface modifiers have been introduced primarily to ease the formation of Ni(III). Experimental evidence suggests that once Ni(III) forms, its reduction back to Ni(II), coupled to hydrogen removal from HMF, can become rate-controlling. If Ni(III) persists too long or reacts too slowly, the surface effectively stalls. This creates a conceptual contradiction for catalyst design: accelerating Ni(II) oxidation alone doesn’t guarantee faster turnover if Ni(III) reduction can’t keep pace and iron complicates this further and when introduced into nickel hydroxide systems, Fe can speed up Ni(III) reduction while simultaneously making Ni(II) harder to oxidize. That dual influence has often been treated as a liability, something to be minimized or avoided. Yet the contradiction is intrinsic to how Fe redistributes electron density around Ni–O units. Ignoring it doesn’t remove it.
A recent research paper published in ACS Catalysis and conducted by Yue Xiao, Ziqi Zhao, Pengfei Long, Jingya Zhang, Dr. Zongyuan Wang, Prof. Jichang Liu, and led by Associate Professor Fuxi Bao from the School of Chemistry and Chemical Engineering, State Key Laboratory Incubation Base for Green Processing of Chemical Engineering at Shihezi University, the researchers developed a Ni(OH)₂/NiFeOxHy hybrid electrode that spatially separates Fe-rich and Ni-rich domains on nickel foam. The structure increases the population of electroactive Ni sites while allowing Fe to accelerate Ni(III) reduction during HMF oxidation. The corrosion step localized Fe within an amorphous NiFeOxHy phase, while electrodeposition placed Ni(OH)₂ on top without forcing extensive Ni–Fe mixing. The investigators relied on that separation to moderate how Fe interacts with the Ni redox cycle.
The team performed microscopy which showed a nanosheet architecture with blurred lattice contrast and frequent crystalline–amorphous junctions. The authors didn’t treat poor crystallinity as a defect and reported that disordered regions and interfaces increase the density of chemically addressable Ni centers, which matters because Fe reduces the fraction of Ni sites that can enter the Ni(III) state. The authors also conducted spectroscopic analysis which supported their view where the Ni sites in the hybrid displayed a higher average valence than those in pure Ni(OH)₂, but Fe showed a corresponding reduction in valence, consistent with interfacial electron redistribution.
Plus, electrochemical measurements clarified how those structural choices play out under reaction conditions. The study examined HMF oxidation currents in alkaline electrolyte and compared the hybrid electrode against Ni(OH)₂ alone and NiFeOxHy alone. The hybrid reached substantially higher anodic currents at comparable potentials, even though Fe-containing systems showed delayed Ni(II) oxidation onset. That delay wasn’t ignored. Instead, the authors emphasized that beyond a certain potential window, the hybrid overtook Fe-free Ni(OH)₂ because Ni(III) reduction proceeded more rapidly. On top of that, product analysis showed near-complete HMF consumption with high selectivity toward 2,5-furandicarboxylic acid. The researchers attributed that selectivity to rapid oxidation of the aldehyde group, which prevents accumulation of reactive intermediates that trigger degradation or polymerization. This interpretation rests on time-resolved concentration profiles not on static yields, and that distinction matters. It’s not just that side products remain low; it’s that intermediates don’t persist long enough to participate in competing chemistry. Shihezi University scientists performed controlled pre-oxidation and reduction experiments to probe the redox mechanism directly and observed that accumulated Ni(III) species disappeared far more quickly in the presence of HMF than in blank electrolyte, confirming that chemical reduction of Ni(III) by HMF is fast. When Fe was present, this reduction accelerated further. However, acceleration came at the cost of fewer Ni(III) sites being generated initially, which is where the hybrid structure becomes essential. Operando vibrational spectroscopy added another layer. Under HMF oxidation conditions, the spectral signatures associated with fully formed nickel oxyhydroxide were suppressed until higher potentials. Instead, transient Ni3+–O species dominated, indicating that Ni(III) intermediates react quickly rather than accumulating. The investigators linked this behavior to a proton-coupled electron transfer process, where Ni3+–O units abstract hydrogen directly from adsorbed HMF. The authors also showed that combining Ni(OH)₂ with NiFeOxHy increases electronic states near the Fermi level and strengthens Ni–O orbital overlap. That electronic structure favors proton capture and electron flow during HMF dehydrogenation. Importantly, the calculations didn’t predict dramatic stabilization of adsorbed HMF alone. They pointed instead to a surface that can handle protons efficiently once adsorption occurs.
To summarize, the new work of Associate Professor Fuxi Bao and colleagues successfully constructed a hybrid electrode by combining a corrosion-grown NiFe oxyhydroxide layer with an electrodeposited Ni(OH)₂ overlayer on nickel foam. Indeed, by separating Fe-rich domains from Ni-rich hydroxide layers, the system tolerates Fe’s suppression of Ni(II) oxidation because the total number of active Ni sites increases. At the same time, Fe’s ability to speed Ni(III) reduction is preserved and even amplified through interfacial coupling. We believe, this has consequences beyond HMF oxidation as many anodic reactions on nickel hydroxides proceed through similar proton-coupled steps involving Ni(III) intermediates. If Ni(III) reduction controls turnover in those systems as well, strategies that focus only on oxidation onset potential are likely incomplete. The present work suggests that controlling the lifetime and reactivity of Ni(III) may be just as important. The new findings also caution against over-interpreting redox peak positions or oxidation currents as direct measures of catalytic competence. A surface that generates Ni(III) easily but can’t reduce it efficiently may look promising in cyclic voltammetry and still perform poorly under steady-state conditions. Downstream applications remain conditional. The hybrid structure depends on maintaining low crystallinity and stable interfaces under prolonged bias. The reported stability tests are encouraging, and long-term restructuring under industrial current densities will be important to conduct in the future research. Lastly, the proposed framework of treating dopants as redox modulators should be considered a useful lens for future catalyst design.
Reference
Xiao, Yue & Zhao, Ziqi & Long, Pengfei & Zhang, Jingya & Wang, Zongyuan & Liu, Jichang & Bao, Fuxi. (2025). Enhancing Selective Electrooxidation of 5-Hydroxymethylfurfural via Coordinating the Contradictory Role of Fe in Ni(II)/Ni(III) Redox Kinetics. ACS Catalysis. 15. 16522-16538. 10.1021/acscatal.5c04226.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.