Significance
Generally, polymers comprise of a molecular structure built up mainly from a large number of similar units bonded together. This material has found many applications since its discovery. In recent times, development of novel polymer membranes for gas separation applications has motivated intense research. This is due, in part, to the fact that gas separation with polymer membranes is energetically more efficient than conventional separation processes. Nevertheless, despite the intensive effort, the landscape of polymers used in the gas industry has not change significantly over the past two decades. This lack of progress in translating results from lab to plant could be attributed to a variety of reasons, but most significantly to the limited knowledge on gas transport in polymers at the molecular level. Attempts to resolve this issue have led to propose a nuclear magnetic resonance (NMR) method that combines multinuclear spectroscopy and pulsed gradient spin echo (PGSE) in order to get a better understanding of the gas transport in polymer membranes at the molecular scale.
Recently, Drs. Leoncio Garrido and Julio Guzmán from the Institute of Polymer Science and Technology (ICTP) at the Higher Council of Scientific Research (CSIC) in Spain presented a study in which they described the findings on the diffusive behavior of pure and mixed carbon dioxide, ethylene, and methane in membranes of a certain glassy polymer (abbreviated as 6FDA-TMPDA), using 1H and 13C PGSE NMR at several pressures and diffusion times. Also, they compare the diffusivity of those gases in the glassy polymer to that in polydimethylsiloxane (PDMS), a rubbery polymer at the temperature at which the measurements were performed. Their work is currently published in the research journal, Macromolecules 2018.
1H and 13C spin−lattice and spin−spin NMR relaxation times and PGSE diffusion measurements were taken by placing polymer membrane strips inside a specially adapted NMR tube at moderately pressurized gases, in a 9.4 T NMR spectrometer.
The authors reported that the relaxation time measurements showed a mono-exponential behavior for all gases, suggesting that either gas molecules were noting a single environment or they were in fast exchange between possible multiple sites. In addition, PGSE NMR diffusion showed that the diffusion coefficients of 13CO2 in the glassy membrane were independent of the diffusion time in all cases studied, similar to the behavior observed in rubbery membranes. In contrast, the diffusive behavior of methane and ethylene in the glassy membrane was found to be dependent on the diffusion time. Moreover, the presence of carbon dioxide and ethylene altered the behavior of methane, reducing the diffusion time dependence and increasing the magnitude of its diffusion coefficient.
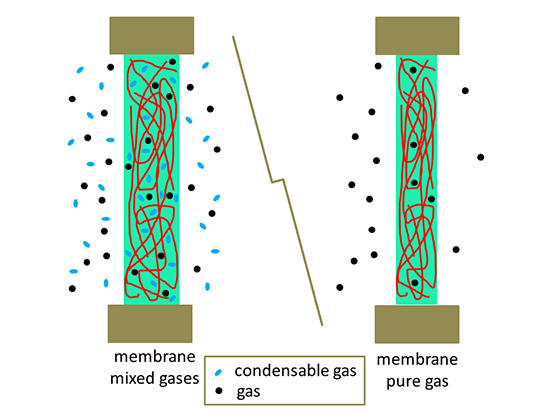
In summary, the study presented an in-depth investigation of the spin−lattice and spin−spin relaxation times and the diffusive behavior of pure and mixed carbon dioxide, methane, and ethylene in glassy membranes of 6FDA-TMPDA using multinuclear spectroscopy and PGSE NMR. A model is proposed to determine the diffusion coefficients of gases exhibiting abnormal (time-dependent) diffusive behavior. Leoncio Garrido and Julio Guzmán finding that the diffusion coefficient of methane increased in the presence of 13CO2 and ethylene, suggests that the sieving properties of the glassy polyimide membranes are compromised even at low concentrations of condensable gases.
Reference
Leoncio Garrido, Julio Guzmán. Influence of Diffusion Time on the Diffusion Coefficients of Gases in Polymers Determined by Pulsed Gradient Spin Echo NMR. Macromolecules 2018, volume 51, page 8681−8688
Go To Macromolecules Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



