Significance
Graphene and its derivatives have taken the scientific community by storm due to their exceptional properties and vast potential for applications in various fields. These remarkable materials have spurred intensive research into manufacturing methods that are robust, scalable, and suitable for creating advanced nanodevices. Recent breakthroughs in the production of graphene-based hybrid composites have showcased their potential in developing switchable devices, sensors, and high-performance nanodevices. Among these composites, graphene oxide (GO) stands out as a promising candidate due to its ability to self-assemble with biomolecules, leading to complex structures with unique functionalities.
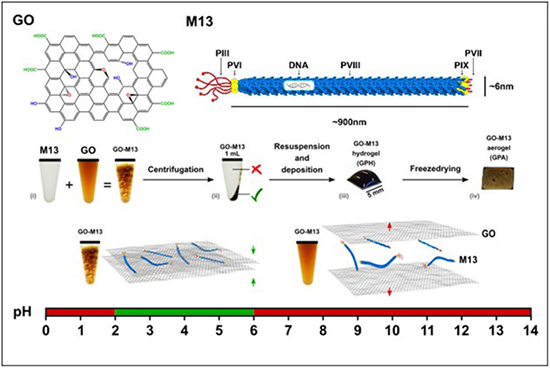
In a new study published in the Journal Nanoscale by Dr. Kate Stokes, Dr. Yiwei Sun, Dr. Paolo Passaretti, Dr. Henry White, and led by Professor Pola Goldberg Oppenheimer at the University of Birmingham explored GraPhage13 aerogels (GPAs), which are micro-porous structures formed through the self-assembly of graphene oxide (GO) and M13 bacteriophage.
GraPhage13 aerogels, also known as GPAs, are intriguing structures that have been engineered through a novel self-assembly process. These aerogels offer a unique combination of properties, including high strength, low density, and a three-dimensional interconnected structure with macroscale dimensions. This combination of characteristics makes GPAs highly appealing for a wide range of applications, from wearable sensors to electrodes and adsorbents. The fabrication of GPAs involves the integration of two key components: graphene oxide (GO) and M13 bacteriophage. M13 bacteriophage is a filamentous virus with unique properties, such as a diameter of 6.6 nm and a length of 880 nm. It replicates by infecting Escherichia coli (E. coli) without destroying the host cell and consists of circular-shaped single-stranded DNA encapsulated by coat proteins. GO, on the other hand, is an atomic layer of sp2-hybridized carbon atoms functionalized with oxygen-containing groups.
The self-assembly of GPAs begins with the integration of GO and M13 at a specific pH, resulting in an aggregate where M13 viral strands act as cross-linkers between the GO sheets. This forms a GraPhage13 hydrogel (GPH), which, when deposited on a supporting substrate in a vacuum, transforms into a stable GraPhage13 aerogel (GPA). The resulting GPA possesses impressive properties, including an ultra-high surface area and low density, making it an excellent candidate for incorporating various nanomaterials like nanoparticles, fluorophores, and polymers.
The authors tried to understand the pH range in which GO and M13 can form a dispersion that facilitates their aggregation for GPA self-assembly. This investigation is essential as it provides insights into the processability of GPAs and their potential applications in environments with varying pH levels. They employed a systematic approach to examine the concentration of ionized groups, the pKa distribution of acid groups, and the zeta potential of GO, M13, GPH, and the combination of GO and M13. The innovative data analysis technique used allowed for a comparison between different experiments, shedding light on the relationship between the ionization of functional groups, surface charge, and pH levels.
The researchers found that GO exhibited a significant variation in the concentration of ionized groups across different pH levels. This variation ranged from 0.75 mmol L−1 mg−1 at pH 2 to 2.15 mmol L−1 mg−1 at pH 11, with a notable increase between pH 5.5 and 7.5. This variation can be attributed to the deprotonation of carboxylic, enolic, and phenolic groups on GO as the pH increased. As the pH increased, the equilibrium shifted, resulting in a higher concentration of negatively charged functional groups.
In contrast, M13 showed a relatively constant concentration of ionized groups (1.89 ± 0.03 mmol L−1 mg−1) across the entire pH range. This stability in ionization is significant as it suggests that M13 maintains its charge in various pH environments. Consequently, the variation in ionized groups observed in GPH and the combination of GO and M13 can be primarily attributed to GO, given M13’s consistent ionization.
The researchers also conducted a detailed analysis of the pKa values of the functional groups present in GO, GPH, and the combination of GO and M13. The pKa values were found to be 3.1, 6.6, and 9.9, corresponding to the ionization of carboxylic and phenolic OH groups in GO. Importantly, no new functional groups were discovered as a result of the interaction between GO and M13. This insight into the pKa values is crucial for understanding the behavior of GO and M13 in different pH environments.
The zeta potential measurements provided further insights into the behavior of GO, M13, and GPH across a wide pH range. GO exhibited the least variation in zeta potential, ranging from -32 ± 1 mV at pH 1.9 to -46 ± 1 mV at pH 11.0. In contrast, M13 displayed a more significant variation, with its zeta potential decreasing from 39.2 ± 0.7 mV at pH 2.1 to -24.6 ± 0.8 mV at pH 5.0 before plateauing between pH 6.1 and 11.0. GPH, interestingly, demonstrated the widest range of zeta potential values, spanning from 31.9 ± 0.1 mV at pH 2.1 to -48.6 ± 0.4 mV at pH 11.1. This wide range indicates the dynamic behavior of GPH in response to changes in pH. The researchers noted that M13 forms stable colloidal dispersions at pH levels below 3 and above 6, but flocculates between pH 3 and 6. The surface charge of M13 is influenced by the protonation and deprotonation of specific residues, making it pH-dependent.
The study’s findings shed light on the pH range in which GO and M13 can aggregate, facilitating GPA self-assembly. It was determined that GO-M13 aggregation occurs between pH 2 and 6. This insight is crucial for understanding the conditions required for GPA fabrication.
Moreover, the research unveiled the origin of GPH’s surface charge, primarily attributed to the M13 bacteriophage. The electrostatic interactions between the carboxylic groups on GO and the positively charged residues of M13, following the protonation of specific residues, play a pivotal role in enabling the self-assembly of GPAs. In conclusion, this innovative investigation, utilizing a novel analytical technique, has provided profound insights into the self-assembly mechanism of GraPhage13 aerogels and the pH range conducive to their fabrication. These insights are pivotal for further research into incorporating various nanomaterials into GPAs, opening up possibilities for the development of graphene-based nanodevices with applications spanning micronano-filters, functional scaffolds, miniaturized sensors, and more.
Reference
Stokes K, Sun Y, Passaretti P, White H, Goldberg Oppenheimer P. Optimisation of GraPhage13 macro-dispersibility via understanding the pH-dependent ionisation during self-assembly: towards the manufacture of graphene-based nanodevices. Nanoscale. 2023;15(32):13304-13312. doi: 10.1039/d3nr00778b.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


