Significance
Adsorptive desulfurization technology is a process used to remove sulfur compounds from various hydrocarbon streams such as gasoline, diesel, jet fuel, and natural gas. This technology is crucial because sulfur compounds, especially in fuels, can lead to harmful emissions when burned, contributing to air pollution and environmental damage.
In a new study published in the peer-reviewed AIchE Journal led by Associate Professor Peng Tan and Professor Lin-Bing Sun and conducted by Min Li, Jun Liu, , Xiao-Qin Liu, from the State Key Laboratory of Materials-Oriented Chemical Engineering, Jiangsu National Synergetic Innovation Center for Advanced Materials (SICAM), College of Chemical Engineering, Nanjing Tech University developed a novel approach to adsorptive desulfurization using silver nanoparticles (Ag(0)) in metal-organic frameworks (MOFs). The researchers embarked on a comprehensive investigation to enhance the efficiency of adsorptive desulfurization, a crucial process in reducing sulfur content in transportation fuels. Their innovative approach combined the fields of material science and chemical engineering, focusing on the use of silver nanoparticles (Ag(0)) within metal-organic frameworks (MOFs) to achieve both efficient sulfur adsorption and energy-effective regeneration. This technique represents a significant leap forward in addressing the limitations of conventional desulfurization methods, which are often hampered by high energy demands and environmental concerns. Their detailed analysis aims to unpack their methodologies, findings, and implications, contributing significantly to the realm of environmental sustainability and pollution reduction.
The first step involved the synthesis of the MOF MIL-101-NO2 through a hydrothermal reaction. This precursor was then transformed into MIL-101-NH2, which served as the base for incorporating AgNO3. The researchers executed this complex process to eventually form Ag@MIL-101-NH2 composites with varied Ag contents. This meticulous preparation was critical in ensuring the composites’ optimal performance in subsequent adsorptive desulfurization processes. To evaluate the photothermal properties of these composites, the team used a xenon lamp with a light filter, simulating solar irradiation. This step was crucial in determining the effectiveness of the composites under light exposure, a key aspect of their innovative desulfurization approach. Infrared thermal imaging and UV-vis spectrophotometry were employed to measure temperature elevations and photothermal effects accurately.
The core of the research centered on adsorption measurements. The team prepared model fuels containing dibenzothiophene (DBT), a common sulfur-containing compound in transportation fuels. They then conducted a series of experiments to assess the adsorption capacity of the Ag@MIL-101-NH2 composites. This included analyzing adsorption isotherms and kinetics, both with and without light irradiation, using gas chromatography to determine sulfur content after treatment.
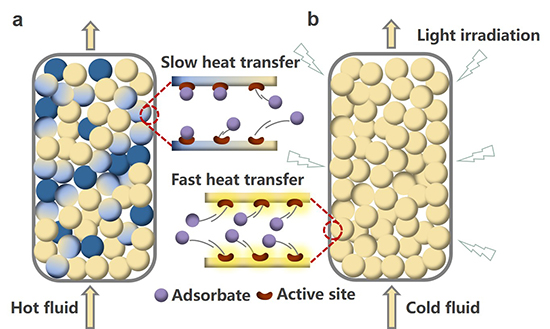
The researchers successfully reported the role of Ag nanoparticles in enhancing adsorption capacity. The Ag nanoparticles, through surface plasmon resonance, were able to convert light into heat effectively. This photothermal effect not only enhanced the adsorption capacity of the composites but also offered a more energy-efficient desorption method compared to traditional techniques. They compared the adsorption capacities of their composites with other adsorbents and conducted competitive adsorption experiments. These tests, which included the presence of other aromatic compounds like toluene, were crucial in evaluating the practical applicability of their composites in real-world scenarios. Moreover, they examined the breakthrough and desorption behavior of the Ag@MIL-101-NH2 composites in a fixed bed setup. This included evaluating the efficiency of photothermal desorption under visible light and comparing it with traditional methods. The team meticulously analyzed the rate of desorption, breakthrough times, and the stability of the composites over multiple cycles.
The researchers calculated the energy efficiency of the photomodulated desorption process and compared it with traditional methods. This analysis was vital in highlighting the potential energy savings and environmental benefits of their approach. The new study represents a major advancement in adsorptive desulfurization technology. By harnessing the unique properties of Ag(0) within MOFs, the researchers achieved a remarkable balance between adsorption efficiency and ease of regeneration. The ability of Ag nanoparticles to rapidly convert light into heat (a process known as surface plasmon resonance) not only enhanced the adsorption capacity but also significantly reduced the energy requirements for desorption. The novel approach of using light to control adsorption and desorption processes in desulfurization represents a paradigm shift in energy efficiency and environmental friendliness. The new technique offers a promising pathway to reduce sulfur emissions from transportation fuels, a crucial step in mitigating air pollution and combating climate change. Furthermore, the study opens new avenues for the application of photothermal phenomena in chemical engineering, potentially revolutionizing processes that require controlled thermal management.
In conclusion Associate Professor Peng Tan and Professor Lin-Bing Sun with their colleagues demonstrated a novel, more efficient, and environmentally friendly approach to adsorptive desulfurization. This study not only contributes to the ongoing efforts to produce cleaner transportation fuels but also opens new pathways for using photothermal phenomena in various chemical engineering applications.
References
Li M, Liu J, Tan P, Liu X-Q, Sun L-B. Photomodulation on adsorptive desulfurization by Ag(0): Photothermal active sites with high stability. AIChE J. 2023; 69(5):e18034. doi:10.1002/aic.18034
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



