Significance Statement
The continued use of fossil fuels, which are non-renewable, has continued to have negative effects of the global climate and economy. Modern technology, for instance, the present day agriculture and irrigation systems, has put a lot of pressure on energy sources. Therefore, the research community has resorted to finding alternative energy sources. They have focused on issues regarding hydrogen storage and have found viable solutions including alcohol fuel cells, such as direct ethanol and methanol fuel cells.
In the recent years, urea has been found as a sustainable hydrogen carrier in view of the large amounts of urea present in wastewater sewage systems due to human and animal waste, and industrial urea production. Above all, urea is nonflammable, relatively non-toxic, stable compound, and solid. Therefore, urea overcomes transportation and storage issues.
Urea in human and animal waste is naturally converted to ammonia and emitted into the environment. Ammonia generates harmful compounds such as nitrates, nitrites, and nitric oxides through oxidation. Therefore, it is important to treat urea in wastewater to prevent health hazards led by urea. A number of methods have been proposed for the decomposition of urea, for instance, chemical oxidation, thermal hydrolysis, and biological decomposition. Unfortunately, these processes demand high active enzymes, high temperatures and/or sophisticated equipment.
A number of materials, such as metal hydroxides, noble and non-noble metals, and metals oxides have been investigated as catalysts for urea oxidation. Researchers have found nickel to be the most suitable for urea electro-oxidation. Unfortunately, nickel catalyst show unsuitable high onset potentials for the oxidation of urea. Therefore, nickel is modified with other metals including zinc, iridium, cobalt, and rhodium. Using multi-metal catalysts unfortunately compromises the available active surface area of the catalyst. Nanosized nickel catalysts therefore seem as promising anodes for direct urea fuel cells.
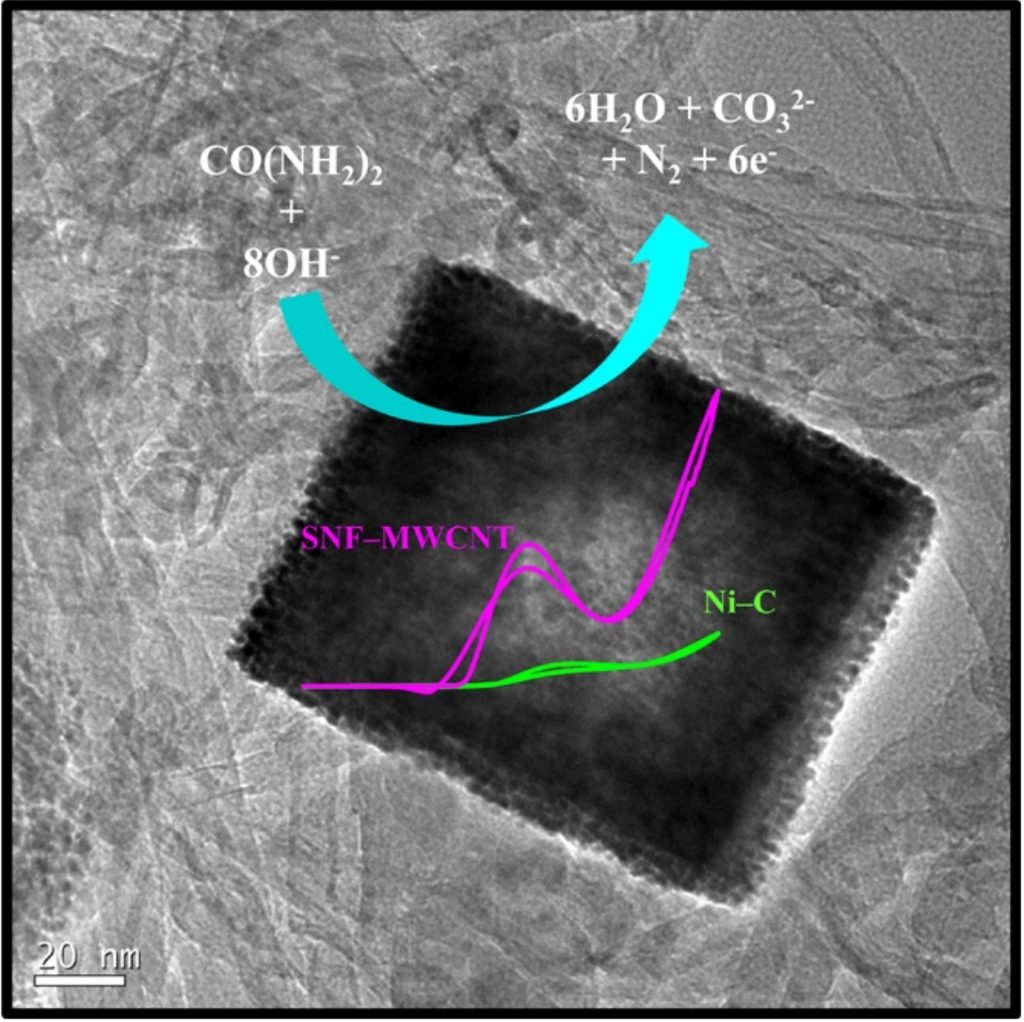
Researchers led by Professor Young Soo Yoon at Gachon University in Republic of Korea in collaboration with Professor Balasubramanian Viswanathan at Indian Institute of Technology Madras synthesized sodium nickel fluoride with Ni2+ and Ni3+ oxidation states deposited on multiwall carbon nanotubes. They reported, for the first time, the synthesis of hollow sodium nickel fluoride nanocubes implementing microwave-assisted method using in-situ template mechanism. Their research work is published in peer-reviewed journal, Electrochimica Acta.
An ionic liquid played a key role in the catalysts preparation. The researchers characterized the physicochemical attributes of the catalyst by X-ray photoelectron spectroscopy, field emission scanning electron microscopy, and X-ray diffraction. They later analyzed the catalysts for urea electro-oxidation implementing electrochemical impedance spectroscopy and chronoamperometry tests.
Sodium nickel fluoride deposited on multiwall carbon nanotubes catalysts showed high catalytic activity as well as stability as opposed to the nickel-multiwall carbon nanotubes without ionic liquid as well as commercial nickel-carbon catalysts.
Using the X-ray photoelectron spectroscopy results, the authors confirmed the interaction between nickel and fluorine and Ni2+/Ni3+ redox pairs with Ni3+ surfaces. Nitul Kakati and colleagues realized an open circuit potential of 0.90 V and a maximum power density of 0.41 mW/cm2 in the direct urea/air fuel cell operated at 55°C with a very low sodium nickel fluoride loading of 2 mg/cm2. The hollow and porous structure of the electrodes composed of Ni3+, and the modification of the electronic architecture of nickel by fluorine presence provided high catalytic performance for the oxidation of urea. Therefore, sodium nickel catalyst is a potential building block for anode catalysts in the direct urea oxidation fuel cells. Quoting Dr. Kakati, “It is now evident that catalyst materials with surface rich in Ni3+ oxidation state is the key to high activity for urea oxidation”.
Reference
Nitul Kakati, Jatindranath Maiti, Kang Soo Lee, Balasubramanian Viswanathan, Young Soo Yoon. Hollow Sodium Nickel Fluoride Nanocubes Deposited MWCNT as an Efficient Electrocatalyst for Urea Oxidation. Electrochimica Acta, volume 240 (2017), pages 175–185.
Go To Electrochimica Acta
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.






