Significance Statement
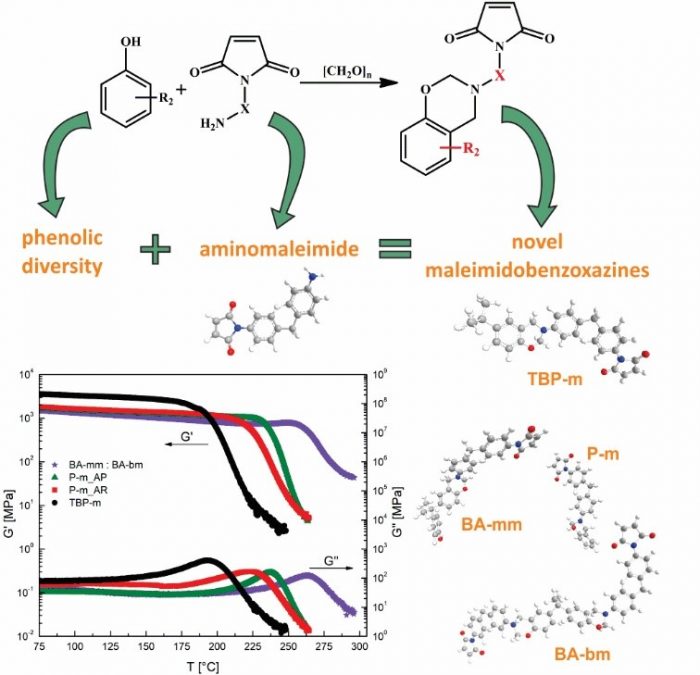
Polybenzoxazines as an advanced material exhibit many eye-catching properties, including: low moisture absorption, good thermal stability, mechanical and electrical properties, high glass transition temperatures, excellent properties in chemical resistance and flame retardant. Moreover, benzoxazine monomers can be acquired from various cheap raw materials leading towards the diverse molecular design flexibility that opens exceptional possibility in further improvements of material properties. The use of maleimide compounds in benzoxazine monomer modifications has been seen to be among the most effective approaches in achieving better polymer properties. According to current literature, there is a vast deficiency of phenolic diversity for current fabricated maleimidobenzoxazine resins.
Matjaž Krajnc and colleagues at the University of Ljubljana in Slovenia, proposed a study on a novel synthetic approach inclined towards greater phenolic diversity of resins by preparing aminomaleimide which can be dissolved with ease in chloroform. They also aimed at the synthesis of versatile maleimidobenzoxazines by incorporating the maleimide molecule directly on the amine reactive compound. Their research work is now published in the peer-reviewed journal, Polymer.
The research team commenced the empirical procedure by preparing the aminomaleimide needed for the preparation of maleimidobenzoxazines on a gram scale without the need for chromatographic purification. The team then prepared maleimidobenzoxazines and compared them with the traditional benzoxazines based on aniline molecule. The new maleimidobenzoxazine monomers were then characterized by, among others, nuclear magnetic resonance analyses, whereas thermal behavior, mechanical properties and thermal stability of cured samples were investigated by differential scanning calorimetry analyses among other techniques. Finally, the use of prepared maleimidobenzoxazines was investigated by the means of Diels-Alder reaction as a possible benzoxazine bearing maleimide group precursor for self-healing purposes.
The authors observed that the novel maleimidobenzoxazines showed similar properties when compared to the ones already presented in the literature thereby showing high glass transition temperature values of over 240°C. Thermo-gravimetric analysis proved improved thermal stability of maleimidobenzoxazines compared to anilinobenzoxazines. Yields of the reaction between aminomaleimide and phenols of up to 95% were achieved in the preparation of maleimidobenzoxazines which was seen as a considerable improvement when compared to previous researches dealing with maleimidobenzoxazine synthesis.
A novel approach for preparation of maleimidobenzoxazines was successfully presented, where for the first time phenols other than 4-hydroxyphenylmaleimide were used in the synthesis of maleimidobenzoxazines leading to the synthesis of various high-performance thermosets. For the first time also, the phenolic diversity in maleimidobenzoxazine compounds has been enabled thus opening many possibilities for tailoring properties of maleimidobenzoxazines through phenolic diversity. In totality, the proposed procedure by the researchers opens promising and novel way that is applicable in the synthesis of diverse maleimidobenzoxazine monomers leading to versatile high performance thermosets.
Reference
Žiga Štirn, Aleš Ručigaj, Matjaž Krajnc. Innovative approach using aminomaleimide for unlocking phenolic diversity in high-performance maleimidobenzoxazine resins. Polymer, volume 120 (2017) pages 129-140.
Go To Polymer
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

