Significance
Macrocycles are large, ring-shaped molecules that consist of twelve or more atoms. These molecules stand out due to their unique structure and the properties they exhibit as a result. For instance, macrocycles have a ring size significantly larger than typical small ring compounds. They might contain various elements, including carbon, oxygen, nitrogen, and sulfur. Their synthesis can be complex, often requiring specialized techniques. This might include multicomponent reactions, ring-closing metathesis, or dynamic combinatorial chemistry. Due to their size and structure, macrocycles often exhibit unique physical and chemical properties. This includes the ability to host other molecules inside their structure, making them useful in supramolecular chemistry. Macrocycles are used as catalysts, in molecular recognition, and in the construction of nanoscale structures. They bridge a gap between small molecule drugs and large biologics, offering unique opportunities in both chemistry and pharmaceuticals. Their ability to interact with complex biological targets and their unique structural features make them an exciting area of research and development in drug discovery and material science. Cyclosporine is a well-known example of a macrocyclic drug used in immunosuppression.
Nanometer distances of molecules in solution refer to the measurement of the spacing or size of molecules at the nanoscale, where one nanometer equals one billionth of a meter. Understanding the nanometer-scale distances between molecules in a solution is key to comprehending their structure and how they interact with each other. This is particularly important in studying proteins, DNA, and other biomolecules, where the specific arrangement and distances between parts of the molecule can determine its function and behavior. In pharmaceutical research, understanding the nanoscale interactions between different molecules, such as a drug and its target, can lead to the development of more effective and targeted therapies. This includes the design of drug delivery systems that can effectively reach their target in the body. Moreover, the distances between molecules at the nanometer scale can influence the rate and outcome of chemical reactions. In solutions, these distances determine how molecules collide and react with each other. Overall, determining nanometer distances of molecules in solution is crucial for advancing our understanding of molecular interactions and structures, which has potential implications in science and technology, from material science to medicine. To this account, a new study published in the Angewandte Chemie International Edition Journal by Hannah Siera, Saber Mehrparvar, Jonas Fax, Mathis Kreuzahler, and led by Professor Gebhard Haberhauer from the University of Duisburg-Essen in Germany, the researchers conducted a detailed study to measure distances within macrocycles in the range of 10 Å using 13C NMR spectroscopy.
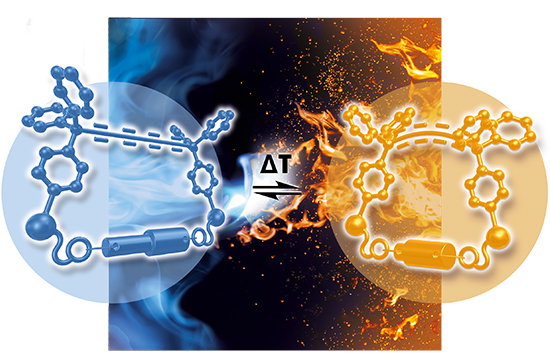
The researchers employed a technique involving 13C NMR Spectroscopy paired with a molecular spring probe. This new methodology allowed for the accurate measurement of distances within macrocycles, particularly in the 10 Å range. By incorporating density functional theory (DFT) calculations and experimenting with various macrocycle structures, the team could observe changes in the chemical shifts of carbon atoms, which correlated with the bending of the molecular spring and thus the distance. The authors presented significant advancements in molecular chemistry, offering a new perspective on the size and shape of molecular systems in solution. The ability to accurately measure distances as small as 10 Å opens up possibilities for better understanding and manipulating molecular structures. Furthermore, the discovery of inverse thermoelasticity at the molecular level offers fascinating opportunities for developing temperature-responsive materials and compounds.
The researchers incorporated a molecular spring, consisting of an oligoalkyne and two trityl groups, into macrocycles. This spring allowed for the measurement of nanometer distances based on the bending of the molecular spring, which in turn affects the chemical shift of the carbon atoms in the 13C NMR spectrum. They used DFT calculations to analyze the relationship between the bending of the molecular spring and the change in the chemical shift of the 13C NMR signals. They optimized geometric parameters of the non-bent and bent states of the molecular spring and calculated the chemical shifts of the sp carbon nuclei. By correlating the distance with the change in chemical shift, a linear relationship could be observed. The authors could subsequently exploit this correlation to determine the distances in different macrocycles experimentally by comparing the chemical shifts of the carbon atoms in these macrocycles with the reference system (molecular spring).
The researchers successfully measured distances within macrocycles in solution, which is challenging due to the presence of multiple conformers and the difficulty in accurately measuring small distances. A significant finding was the observation of inverse thermoelasticity at the molecular level. They found that the macrocycles contracted upon heating, a phenomenon contrary to typical thermoelastic behavior in macroscopic systems. The new method allows for a detailed understanding of molecular structures in solution and opens new possibilities for manipulating these structures based on temperature changes. In conclusion, the findings of Professor Gebhard Haberhauer and associates have potential applications in designing temperature-responsive materials and understanding molecular interactions in various fields, including drug design and nanotechnology. Moreover, the combination of DFT calculations and experimental NMR data provided a robust and reliable way to measure distances in molecular systems, overcoming previous limitations in this area of research.
Reference
Siera H, Mehrparvar S, Fax J, Kreuzahler M, Haberhauer G. Measurement of Nanometer Distances in Solution via 13 C NMR Spectroscopy-Shrinking of Macrocycles with Increasing Temperature. Angew Chem Int Ed Engl. 2023;62(25):e202301465. doi: 10.1002/anie.202301465.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



