Environmental response regulation via chemical functionality and end groups placement
Significance
Stimuli-responsive polymers have been extensively researched for potential biomedical applications owing to their ability to self-assemble in various well-defined nanoscale morphologies in aqueous media in response to different external stimuli. There are two main categories of stimuli-responsive polymers, namely, temperature-responsive polymers and pH-responsive polymers. Temperature-responsive polymers, such as PNIPAM-based polymers, are sensitive to changes in solution temperature. They include those that become hydrophobic when the solution’s temperature exceeds a critical value presenting lower critical solution temperature (LCST and those that become hydrophilic when the temperature exceeds a critical value representing upper critical solution temperature (UCST). On the other hand, pH-responsive polymers such as PDMAEA-based polymers change their conformational and hydration state in response to changes in the solution pH. Nevertheless, the synthesis of block copolymers comprising of both thermo- and pH-responsive polymers have not been fully explored, despite its potential for polymer nanostructure manipulation via external environmental triggering and biomedical applications.
Presently, the reversible addition-fragmentation chain transfer polymerization method is widely used to synthesize various well-defined stimuli-responsive block copolymers due to its versatility, controllability, and ability to incorporate a wide variety of functional monomers. To this account, Dr. Stergios Pispas and his PhD graduate Despoina Giaouzi from the National Hellenic Research Foundation in Greece synthesized a series of doubly temperature- and pH-responsive diblock copolymers (poly(N-isopropylacrylamide)-b-poly(2-(dimethylamino)ethyl acrylate (PNIPAM-b-PDMAEA) via sequential reversible addition-fragmentation chain transfer polymerization method. The main aim was to investigate the effects of temperature and pH on the self-assembly properties of the synthesized copolymers in aqueous media. Their work is currently published in the European Polymer Journal.
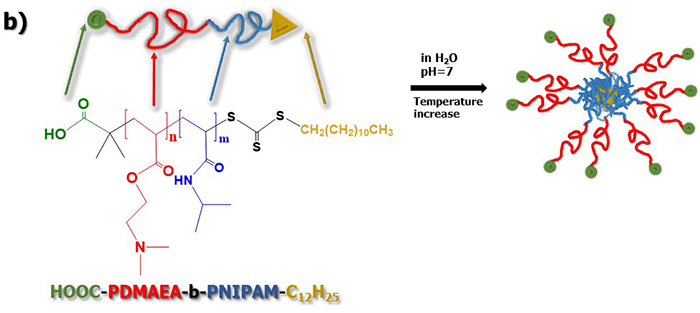
In their approach, hydrophobic C12H15 and hydrophilic COOH groups were selectively placed at the chain end of either thermo- or pH-responsive block, and their effects on the self-assembly behavior of the diblock copolymers examined. The tertiary amine groups of PDMAEA blocks were quaternized with methyl iodide to convert amine precursor diblocks to strong cationic block polyelectrolytes. The self-assembly behavior of the amine-based and quaternized diblock copolymers were investigated in aqueous media of varying temperature and pH using a combination of light scattering and fluorescence spectroscopy techniques.
Results showed that the self-assembly behavior of the novel diblock copolymers was affected by both pH and temperature of the aqueous media solution. The copolymer formed loose aggregate structures at room temperature attributed to the existence of the hydrophobic C12H25 functional groups. At temperature above the lower critical solution temperature of the PNIPAM block, the formation of nano-assembled structures with PNIPAM and PDMAEA blocks occupying the inner and outer parts of the formed aggregates respectively, was observed. On the other hand, the effects of the solution pH were mainly attributed to the presence of PDMAEA block. Molecular characterizations revealed that the composition and molecular weights of the synthesized PNIPAM-b-PDMAEA block copolymers could be effectively controlled and determined to a large extend the physicochemical properties of the copolymer in aqueous solutions.
In summary, the study is the first to successfully synthesize diblock copolymers consisting of one thermo-responsive block (PNIPAM) and one pH-responsive block (PDMAEA) with different compositions, molecular weights and end group placements. The results showed that the self-assembly behaviors and aqueous solution properties could be manipulated by varying the environment stimuli such as pH and temperature as well as the composition and structural architecture of the diblock copolymers. In a statement to Advances in Engineering, Dr. Stergios Pispas, Director of Research at the National Hellenic Research Foundation stated that “block chemical nature, macromolecular architecture, including end group hydrophobicity/hydrophilicity and position, as well as the overall molecular properties of the synthesized diblock copolymers make them promising candidates for biomedical applications, especially in the fields of drugs and gene delivery”.

Reference
Giaouzi, D., & Pispas, S. (2020). PNIPAM-b-PDMAEA double stimuli responsive copolymers: Effects of composition, end groups and chemical modification on solution self-assembly. European Polymer Journal, 135, 109867.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.

