Significance
The need to reduce harmful gases emission in the atmosphere is currently a great concern among the scientists and policymakers. As such, various industrial processes such as adsorption, extractive distillation, and hydrodesulfurization have been developed in the removal of sulfur from liquid fuels or chemical products. In case of desulfurization of gasoline products, despite the high efficiency of the hydrodesulfurization process, it is associated with loss of octane number. This has led to the advancement of the technology and introduction of a blending component methyl tert-butyl ether (MTBE). Owing to its high-octane number, it is capable of producing lower-sulfur gasoline blends with high octane number. Therefore, removing sulfide from MTBE is increasingly becoming a challenge in well controlling the sulfur content of liquid fuels. Researchers have been looking for better techniques for desulfurization of MTBE and have identified extractive distillation as a promising separation method.
Researchers at East China University of Science led by Professor Hui Sun developed an extractive distillation method for desulfurization of MTBE. They used dimethyl disulfide (DMDS) as the model compound. They purposed to significantly minimize the sulfur content of the products. Their work is published in the journal, Industrial and Engineering Chemistry Research.
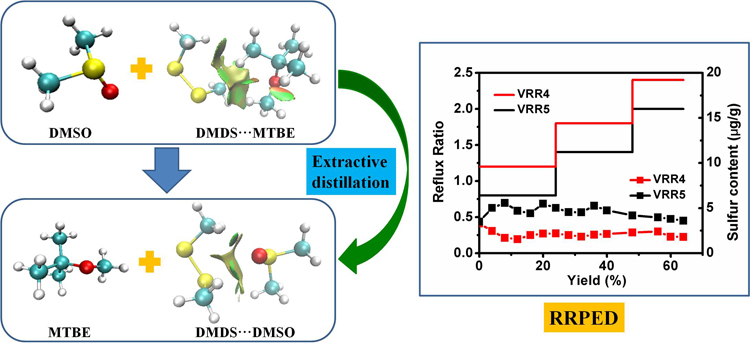
Briefly, the authors commenced their experiment by defining and mapping the intermolecular interactions among the solvents DMDS and MTBE using the reduced density gradient (RDG) and atoms in molecules (AIM) analysis methods. On the other hand, density functional theory and dissipative particle dynamics were employed in the calculation of the distribution of the DMDS and the interaction energies in the solvents. Furthermore, they developed a reflux-ratio-programmed batch extractive distillation (RRPED) process to assist in achieving the desirable high desulfurization efficiency.
Guo-Xiong Zhan and colleagues observed that the interactions between the DMDS and the solvents were van der Waals and weak hydrogen bond forces. Also, according to the computational results for the distribution of DMDS in the solvents and MTBE, dimethyl sulfoxide exhibited the highest efficiency for the DMDS removal due to its strong interaction with DMDS as compared to its counterparts. Consequently, the authors recorded the highest reduction of the sulfur content from 2000 ug/g to less than 5 ug/g. This was attributed to the enhanced desulfurization efficiency using the developed RRPED process.
The East China University of Science and Technology researchers has successfully developed a novel RRPED process for the removal of DMDS from MTBE. By overcoming the challenges posed by the previous techniques, they obtained a high desulfurization efficiency. Suitable operation conditions must be set for the RRPED process to ensure proper functionality. The flux ratio determined both the economy and general performance of the extractive distillation process.
Among the three solvents used, dimethyl sulfoxide (DMSO) was considered the most suitable solvent for this process due to its high desulfurization efficiency. The feasibility of the developed process was confirmed by the similarities in the results obtained for both the simulation and experimental cases. Owing to its efficiency and relatively low energy consumption, the study will advance several industrial processes for removal of sulfur from liquid fuels or chemical products.
Reference
Zhan, G., Shen, B., Sun, H., & Chen, X. (2018). Extractive Distillation Approach to the Removal of Dimethyl Disulfide from Methyl Tert-Butyl Ether: Combined Computational Solvent Screening and Experimental Process Investigation. . Industrial & Engineering Chemistry Research, 57(9), 3348-3358.
Go To Industrial & Engineering Chemistry Research Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



