Significance Statement
Nitrogen is an important building block for almost all forms of life considering that it is needed to biosynthesis basic components of plants and other living organisms. Living organisms can consume nitrogen in usable forms that are obtained by chemical reaction with hydrogen and oxygen. For this reason, nitrogen compounds can be found in amino acids, nucleic acids, plant cells, and proteins.
Nitrogen is in abundant supply in the atmosphere; about 78.08% of air is composed of nitrogen. Unfortunately, this abundant nitrogen source is not available to a good number of living organisms in view of the extreme difficulty in breaking its triple bond as well as its stable electronic configuration. Therefore, this makes the initial reaction step of the conversion energy demanding.
Nitrogen fixation that converts molecular nitrogen into simple compounds including ammonia and nitric oxide can be used further for the preparation of more complex molecules. Unfortunately, this is the most challenging step of nitrogen utilization by living organisms. The Haber-Bosch process, binding nitrogen with hydrogen to produce ammonia at high pressure, is perhaps the most efficient process for fertilizer production. However, industrial ammonia preparation is the most energy intensive chemical process.
For this reason, there is a need to come up with sustainable processes for nitrogen fixation. Several alternatives have been investigated including biological nitrogen fixation and nitrogen fixation with metallocomplex homogeneous catalysts under ambient pressure. Plasma technology, and particularly gliding arc plasma, is a promising candidate in this industry. However, much is still unknown regarding the underlying mechanism.
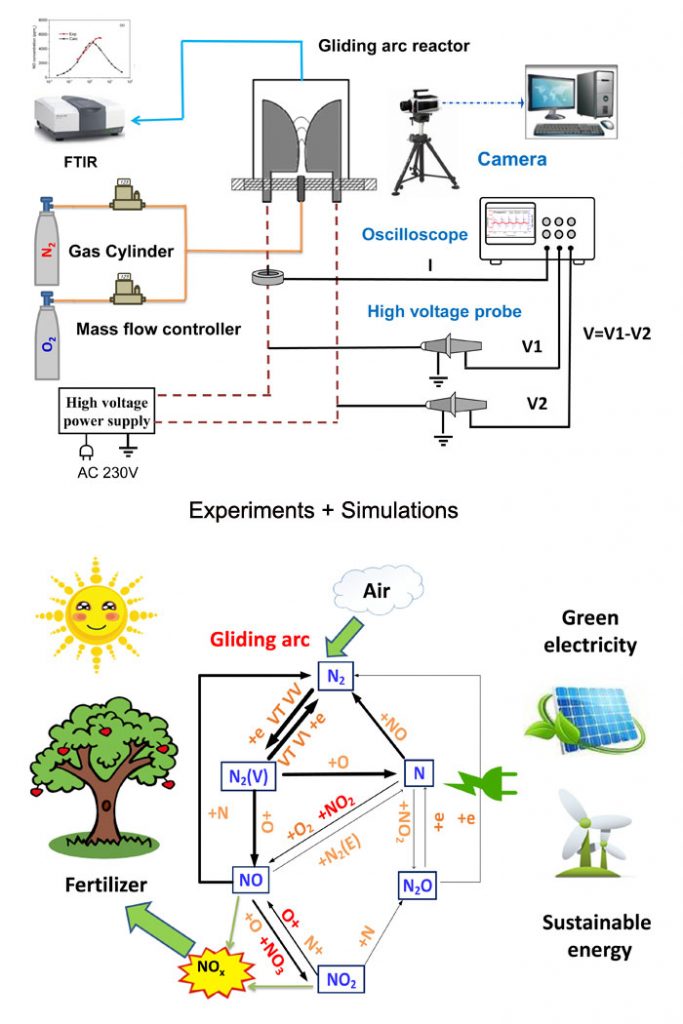
Belgian researchers at University of Antwerp, Weizong Wang, Stjin Heijkers and Annemie Bogaerts, in collaboration with Bhaskar Patil and Volker Hessel at Eindhoven University of Technology, studied the NOx production in a pulsed-power gliding-arc plasma reactor by a chemical kinetic model. They performed experiments to benchmark the model, and reasonable agreement was reached between the computed and measured NO and NO2 yields. Their research work is published in ChemSusChem (with the highlight of very important paper) and it was also featured as cover story of the journal.
The research team wanted to get an in-depth understanding of nitrogen fixation via NOx production in gliding arc plasma by means of combined experiments and a zero-dimensional kinetics model. They compared their experimental outcomes with the predictions of the model and found good agreement for NO, NO2 and the total NOx production, NO2 and NO selectivity, energy consumption, and energy efficiency for the whole range of N2/O2 ratios in the mixture. This indicated that the proposed model could be implemented to elucidate the most dominant reaction pathways for the production of NOx.
Their study also revealed that vibrational excitation of nitrogen could help to overcome the reaction energy barrier of the non-thermal Zeldovich mechanism, and could therefore improve NO production. This provided an energy efficient approach for NO formation in the gliding arc. The outcomes of the study also indicated that the most important reaction for nitrogen oxide formation was the oxidation of NO by oxygen atoms.
The researchers also compared their experimental results with those of thermal NOx production. The NOx yield and energy efficiency obtained in the gliding arc were much higher than the thermal values, due to the non-equilibrium properties of the plasma, since the chemistry of the conversion is initiated by energetic electrons.. Although the energy consumption achieved is still higher than for the Haber-Bosch process, indicating that further improvements are necessary, the authors believe that the gliding arc is a promising building block for industrial-scale nitrogen fixation.
The authors believe the new plasma process open doors to new windows of opportunity concerning distributed fertilizer manufacturing. This had a social dimension as this is needed mostly in less developed regions with large transport distances such as in Africa. Thus IT contributes to the megatrend resources (hunger). Hunger is given despite having Haber-Bosch.
There are a lot of applications for this technology. For example, farmers in remote areas will be able to create mineral fertilizers on site with wind or solar energy as a sustainable alternative to current conventional fertilizers. This certainly opens up opportunities in developing countries and regions where renewable energy is currently underused.
Reference
Weizong Wang, Bhaskar Patil, Stjin Heijkers, Volker Hessel, and Annemie Bogaerts. Nitrogen Fixation by Gliding Arc Plasma: Better Insight by Chemical Kinetics Modelling. ChemSusChem 2017, 10, 2145 – 2157.
Go To ChemSusChem
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




