Significance
The escalating environmental burden of conventional petroleum-derived plastics has driven the scientific community to seek biodegradable, renewable polymeric alternatives. Over the past decades, polyesters have gained prominence as potential eco-friendly replacements due to their inherent degradability and compatibility with biological systems. Yet, despite advances in sustainable polymer chemistry, challenges persist in achieving structural diversity and functional tunability without compromising synthetic efficiency or environmental safety. A key strategy to overcome these challenges lies in developing renewable monomers that permit post-polymerization modification, thereby allowing versatile molecular architectures from a single synthetic framework. However, the incorporation of reactive sites directly into the polymer backbone—without resorting to multi-step protection or modification schemes—remains a formidable task.
Among the various monomer classes explored, epoxide–cyclic anhydride copolymerization has emerged as a particularly efficient route for generating aliphatic polyesters under mild catalytic conditions. The method is attractive not only for its atom economy but also for its compatibility with a wide range of catalytic systems. Yet, the limited availability of renewable, functionally diverse epoxides constrains the method’s broader applicability. Functional handles such as alkenes have been introduced before, enabling post-polymerization chemistry, but aldehyde-bearing monomers—which offer unique chemical reactivity and open possibilities for imine, acetal, or cross-linking reactions—remain scarce.
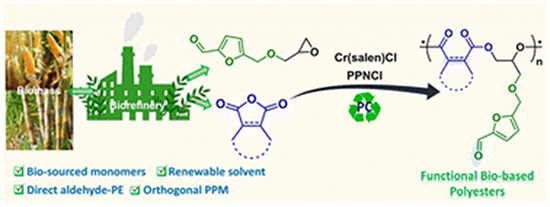
To this account, new research paper published in Polymer Chemistry and conducted by Dr. Mani Sengoden, Dr. Vanaparthi Satheesh, Dr. Sriparna Sarkara and led by Professor Donald J. Darensbourg from the Texas A&M University, the team developed two complementary polymer systems: aldehyde-functionalized polyesters obtained from the copolymerization of the 5-hydroxymethylfurfural-derived epoxide (CFGE) with various cyclic anhydrides, and their orthogonally modified derivatives featuring both imine and thioether functionalities. These models demonstrate selective, independent reactivity of aldehyde and alkene groups within the same polymer chain. Their approach combines sustainable monomer synthesis, green catalysis, and precise post-polymer modification, offering a powerful route to customizable, biobased polyesters with tunable physical and chemical properties
The research commenced with the preparation of the 5-hydroxymethylfurfural-derived epoxide monomer, CFGE, followed by its copolymerization with a variety of cyclic anhydrides using the Cr(salen)Cl/PPNCl binary catalyst. The reaction parameters were carefully optimized to balance conversion, selectivity, and sustainability. Initial trials conducted in toluene at 90 °C afforded nearly complete conversion to poly(CFGE-alt-PA) within one hour, confirming the high catalytic activity of the chromium complex. Seeking a greener alternative, the team replaced toluene with propylene carbonate, achieving full conversion within 18 hours at 90 °C, and within a single hour upon raising the temperature to 120 °C. Control reactions using either component of the catalytic pair independently led to significantly lower conversions, highlighting the synergistic function of the Cr(salen)Cl–PPNCl system.
Subsequent experiments expanded the substrate scope by copolymerizing CFGE with diverse bio-based anhydrides, including phthalic, maleic, succinic, glutaric, cyclohexene, and norbornene anhydrides. The reactions consistently yielded alternating copolyesters with high selectivity. Characterization via FT-IR, ¹H NMR, and ¹³C NMR confirmed the formation of ester linkages and alternating structures. Thermal analyses revealed degradation temperatures (Td₅%) between 190 °C and 300 °C and glass transition temperatures (Tg) ranging from −12.4 °C to 40.1 °C, depending on the rigidity of the cyclic anhydride. The polyesters derived from more rigid anhydrides, such as exo-norbornene, exhibited higher Tg values, reflecting the direct relationship between monomer structure and polymer thermal behavior. One of the most intriguing aspects of the study was the controlled stereochemical behavior observed during copolymerization with exo- and endo-norbornene anhydrides. NMR and 2D NOESY analyses demonstrated that the original cis-2,3-(exo,exo) and cis-2,3-(endo,endo) configurations were retained, confirming that no isomerization occurred under the employed conditions—an observation that contrasts with previous studies where structural rearrangements were typical. The presence of pendant aldehyde groups in the resulting polyesters provided versatile functional handles for post-polymerization modification. Reactions with primary amines yielded corresponding imines, evidenced by the disappearance of the aldehyde carbonyl stretch in FT-IR spectra and the emergence of characteristic imine signals in ¹H NMR. Further, thiol-ene reactions were selectively carried out at the norbornene double bonds using 1-butanethiol under AIBN-catalyzed conditions. The orthogonality of these transformations was striking: while the alkene groups reacted quantitatively, the aldehyde moieties remained intact. Remarkably, a one-pot dual PPM was demonstrated by treating poly(CFGE-alt-exo-NA) simultaneously with sec-butylamine and 1-butanethiol, producing both imine and thioether functionalities without cross-reactivity. The molecular weight increased from 5.26 kg mol⁻¹ to 8.48 kg mol⁻¹, confirming successful double functionalization and highlighting the controllable chemistry accessible through this renewable polymer platform.
In conclusion, Professor Donald J. Darensbourg and colleagues introduced a transformative route to biobased functional polyesters, coupling renewable monomer design with selective orthogonal post-modification. By employing a hydroxymethylfurfural-derived epoxide in combination with cyclic anhydrides, the Darensbourg group effectively bridged the gap between green chemistry and advanced polymer functionality. The dual presence of aldehyde and alkene moieties within the polyester backbone allows for unprecedented modularity—properties rarely accessible in bio-based materials. Such orthogonal reactivity not only expands the synthetic versatility of sustainable polymers but also lays the foundation for multifunctional materials tailored to biomedical, coating, and responsive system applications. The strategic use of propylene carbonate as a solvent underscores the group’s commitment to environmentally responsible synthesis. This shift from volatile organic solvents to a benign, recyclable medium exemplifies the principle of designing safer chemical processes. Furthermore, the demonstration that the Cr(salen)Cl/PPNCl catalytic system maintains efficiency under these greener conditions reinforces its practical value for scalable and sustainable operations. The ability to achieve high conversions with renewable substrates suggests the potential for industrial adaptation, particularly in the production of degradable specialty polymers. Beyond the immediate synthetic advances, the orthogonal post-polymerization modification strategy represents a conceptual leap in macromolecular design. The selective transformation of aldehyde groups to imines—coupled with independent thiol-ene addition on alkenes—allows dual functionalization in a single pot, enabling property tuning without polymer degradation or unwanted cross-linking. This modularity could enable the creation of responsive hydrogels, adhesive coatings, or degradable drug delivery matrices whose chemical composition can be tailored at will. Moreover, the successful retention of stereochemical integrity during polymerization highlights a mechanistically clean process that preserves structural fidelity, a key requirement for consistent performance in applied materials.
At a broader level, the study aligns with the global shift toward circular materials design, where renewable feedstocks, controlled reactivity, and degradability converge to form a sustainable polymer economy. The work exemplifies how rational molecular engineering—anchored in fundamental catalysis—can transform an abundant biomass derivative into a flexible chemical platform for modern polymer science. By revealing how aldehyde and alkene functionalities can coexist and be independently manipulated, the authors provided a blueprint for developing next-generation biopolymers that are not only green but also functionally rich.
Reference
Sengoden, Mani & Satheesh, Vanaparthi & Sarkar, Sriparna & Darensbourg, Donald. (2025). Renewable hydroxymethylfurfural epoxide and cyclic anhydride copolymerization: a green route to functional biobased polyesters. Polymer Chemistry. 16. 10.1039/D5PY00800J.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


