Significance
Photochemistry concerns the chemical effects of light; more so, the chemical reaction caused by absorption of ultraviolet, visible light and infrared radiation. In particular, visible light-driven photochemical methods have proven to be valuable tools that enable achieve various challenging organic transformations. In fact, it is well documented that a variety of metal complexes, organic dyes, and inorganic semiconductors exhibit efficiency in photoredox reactions through visible light excitation. However, a single visible photon (≤300 kJ mol-1) does not provide sufficient energy for the activation or dissociation of many strong chemical bonds such as aryl-Br, aryl-Cl, C-O, and C-H bonds.
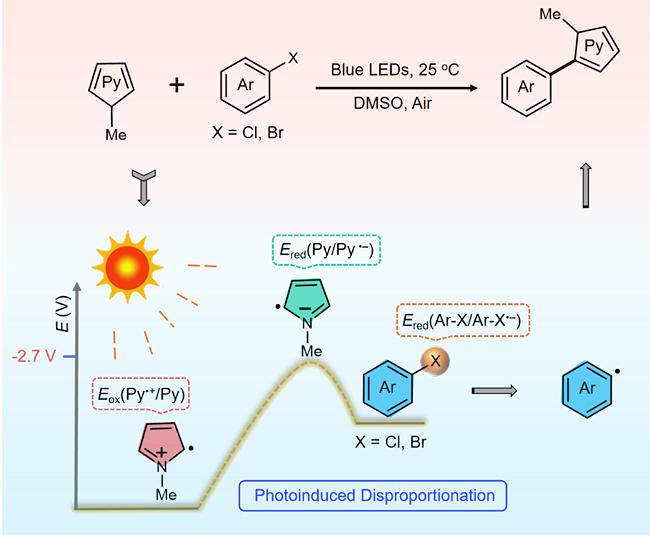
Pyrrole and its derivatives are an important class of heterocyclic compounds, containing pyrrole rings, which are widely distributed in many biologically active natural products and pharmaceuticals. Literature has it that besides the extensive biological and organic synthetic interest in pyrrole derivatives, polymers derived from pyrrole and its derivatives are capable of photochemical polymerization via photoinduced disproportionation processes under visible light illumination. Despite extensive research studies in photochemical polymerization of pyrrole and its derivatives, the application of radical-anion species of pyrrole during C-H activation using visible light has less unexplored.
To bridge this gap, researchers from the Department of Chemistry at Syracuse University in New York: Dr. Zhi-Jun Li, Elan Hofman, Andrew Hunter Davis and Professor Weiwei Zheng, in collaboration with Shuya Li and Professor Gyu Leem from the Department of Chemistry at the State University of New York College of Environmental Science and Forestry investigated that pyrrole derivatives are possibly responsible for the initiation of aryl bromide and chloride reduction, specifically, the photoreduction of aryl halides through a detailed study of alkyl halide arylation with pyrrole derivatives in the absence of any photocatalysts or additives. Their work is currently published in the research journal, Green Chemistry.
The research team proposed a photoinduced disproportionation approach without the addition of any photocatalysts or additives to afford radical anions of pyrrole derivatives, which have enough reduction power to transfer an electron to aryl halide, giving rise to the corresponding aryl radical to afford the desired C–H arylated heterocyclic product. They demonstrated that the photoinduced disproportionation process of pyrrole derivatives led to radical anion pyrrole derivatives, which acted as strong reduction species to undergo single electron transfer (SET) to an aryl halide, producing an aryl radical after carbon–halogen bond cleavage. Subsequently, the capture of aryl radicals by pyrrole derivatives produced C–H arylated heterocyclic products. The team further noted that products generated in situ open up the possibility to follow the photoinduced disproportionation pathway as well, to transfer electrons to aryl halides, thereby accelerating the conversion.
“This new approach provides a new radical initiation mode using aryl halides through a photochemical approach that is highly desirable in organic synthesis, materials, and life sciences.” Zheng told Advances in Engineering.
In summary, the study by Professor Weiwei Zheng and colleagues presented a novel radical initiation mechanism via photoinduced disproportionation approach for the photoreduction of relatively inactive aryl bromides and chlorides. The proposed photoinduced disproportionation pathway offers new photocatalytic insights to show that the energy absorbed from visible light allows to drive transformations that were traditionally believed to be challenging, while also enriching the understanding of the photoreduction mechanism of many relevant reactions.
Reference
Zhi-Jun Li, Shuya Li, Elan Hofman, Andrew Hunter Davis, Gyu Leem, Weiwei Zheng. Visible-light induced disproportionation of pyrrole derivatives for photocatalyst-free aryl halides reduction. Green Chemistry, 2020, volume 22,1911.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




