Significance Statement
Due to the considerable increase of concentration of nitrous oxide in the atmosphere resulting mostly from anthropological effects, scientists have faced the challenges of lowering the effects of nitrous oxide on environment as it is regarded as a greenhouse gas contributing to global warming and also ozone layer depletion.
Decomposing nitrous oxide by a photocatalytical process under ultraviolet irradiation can certainly offer solutions to this issue. A semiconductor such as titanium dioxide shows photocatalytic activity and can be used as a photocatalyst. However, its large band gap and quick electron-hole recombination limits its photoactivity.
Another semiconductor, graphitic carbon nitride also exhibits photocatalytic activity. They have also been studied to provide support as photocatalytic materials. However, their morphological property and abrupt electron-hole recombination limits their potential in photocatalytical processes. In order to limit the electron-hole recombination phenomenon which occurs in both discussed photocatalysts, generation of heterojunction in the semiconductor or changes in their morphologies could serve as a means for achieving this.
In a recent article published in Applied Surface Science, Kamila Koči and colleagues prepared different mixing ratios of titania and graphitic carbon nitride photocatalyst for decomposition of nitrous oxide under ultraviolet irradiation at wavelengths of 254 nm (UVC) and 365 nm (UVA).
They prepared mixing ratios of 1:2, 1:4 and 1:6 with respect to titania and graphitic carbon nitride photocatalyst nanocomposite by means of mechanical mixing in water suspension before the calcination took place. All fabricated nanocomposites were characterized by several experimental techniques and their decomposition effect on nitrous oxide under photocatalyst was observed with an 8 W Hg irradiation lamp.
Results from all the characterization techniques show the successful deposition of titania on the graphitic carbon nitride surface for all the prepared nanocomposite photocatalysts following the generation of heterojunction in all of them.
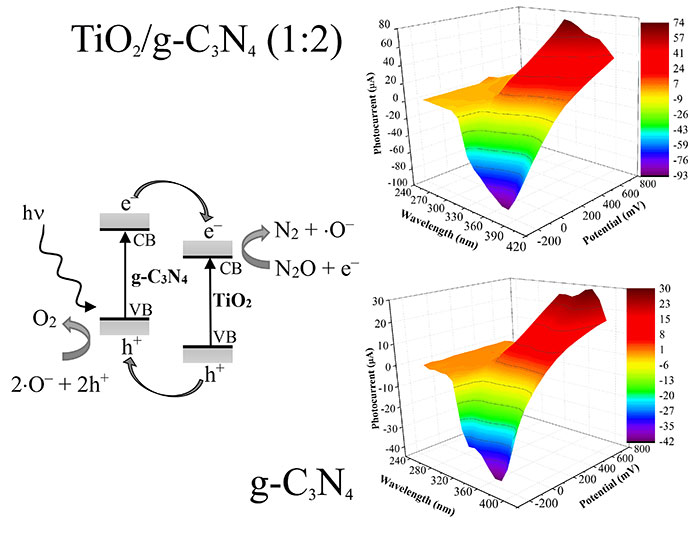
Their photocatalytic decomposition rate on nitrous oxide was also investigated for 14 hours. They discovered all the tested nanocomposites had comparable photocatalytic activity under ultraviolet irradiation at wavelength of 254 nm. However, under wavelength of 365 nm, the highest decomposition rate for nitrous oxide conversion was observed with titania and graphitic carbon nitride in mixing ratio 1 to 2.The decomposition process at both wavelengths increased as the time progressed.
All photocatalyst nanocomposites were compared with a commercial photocatalyst, Evonik P25 and exhibited higher photocatalytic activity. They also observed, with the aid of kinetic constants evaluation, that increase in proportion of graphitic carbon nitride resulted in decrease in photocatalytic activity. At longer wavelength, the composite titania/graphitic carbon nitride in ratio of 1:2 had the highest photocurrent generation as a result of the created heterojunction, which limits the electron-hole recombination phenomenon.
The research team was able to develop a photocatalyst which converts one of the potential greenhouse gases, nitrous oxide into non-hazardous gases of nitrogen and oxygen respectively.
Reference
Kočí, K., Reli, M., Troppová, I., Šihor, M., Kupková, J., Kustrowski, P., Praus, P. Photocatalytic Decomposition of N2O over TiO2/g-C3N4 Photocatalysts Heterojunction, Applied Surface Science 396 (2017) 1685–1695.
Go To Applied Surface Science
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.








