Significance Statement
United States ethylene production has shown a rapid capacity growth recently due to the shale gas boom. As ethylene production is one of the largest contributors to the energy consumption and greenhouse gas emissions within the chemical industry, the expansion of ethylene capacity is likely to dramatically increase the U.S. energy consumption and greenhouse gas emissions. Such increase could be mitigated by the adoption of state-of-the-art and emerging technologies, which drives the development of alternative pathways and advanced technologies for improving energy efficiency and reducing greenhouse gas emissions.
In a recent article published in journal, Industrial & Engineering Chemistry Research, Yao et al. (2016) developed a bottom-up model to evaluate the potential changes of primary energy consumption and greenhouse gas emissions in the U.S. ethylene production associated with different technologies pathways in a prospective fashion.
Previous studies evaluating the potential of reducing energy use and greenhouse gas emissions associated with the ethylene industry are not specifically for the United States, and most of them do not include prospective estimation for the future. In addition, most of these studies focused on direct energy use and emissions without broader consideration of the indirect (or upstream) energy use and greenhouse gas emission associated with the extraction refining and distribution of ethylene plant feedstock.
The bottom-up model developed in this research includes three modules: a cradle-to-gate energy and emission analysis module, a technology assessment module, and a prospective analysis module. The cradle-to-gate energy and emission analysis module is a first-principle unit-process system model that estimates the primary energy consumption and greenhouse gas emissions of three feedstock pathways: shale gas to ethylene, conventional natural gas to ethylene and naphtha to ethylene.
Integrated with the cradle-to-gate energy and emission analysis module, technology assessment module estimates the energy consumption and greenhouse gas emission intensities of state-of-the-art technology and five emerging technologies, including catalyst-assisted production, microwave-enhanced cracking, micro-channel process technology, membrane and distillation hybrid process, and holler fiber technology. Emerging technologies are adopted in existing plants as retrofit options and in new plants as addition to state-of-the-art processes. The variances and uncertainties of system parameters were taken into consideration, and Monto Carlo simulation was used to estimate the ranges of cradle-to-gate energy and greenhouse gas emission intensities of ethylene production by different technologies. Based on the result, emerging and state-of-the-art technologies showed reductions of primary energy and greenhouse gas emission intensities when compared with conventional steam cracking process. The intensity results for conventional ethylene production by different feedstocks and technology pathways were used in the prospective analysis module for projecting annual cradle-to-gate primary energy and greenhouse gas emissions of United States ethylene production from 2015 to 2040.
Based on the results, given current practices and technologies, the primary energy consumption and greenhouse gas emissions of the U.S. ethylene industry will greatly increase. The impact can be mitigated by adopting the state-of-the-art technology, but the mitigation is not large enough to cancel out the increase of energy consumption led by capacity growth. The adoption of emerging technologies can further reduce energy and greenhouse gas emissions, however, the reduction potential is subject to the uncertainty of technology performance. The authors suggest the inclusion of reducing uncertainty in future R&D work to enable more robust prospective analysis. Among five emerging technologies, catalyst-assisted production and microwave-enhanced cracking technologies applied to steam cracker tend to have larger savings than the other three technologies. The results also show that in the short term, catalyst-assisted production technology is more favorable regarding energy reduction, however, in a long-term, microwave enhanced cracking may have greater potential especially given that it is powered by electricity and thus can be integrated with renewable power generation in the future. The selection of technology should be considered on a case-by-case basis as the appropriateness and potential of each technology depends on the reduction target (e.g., short-term or long-term), resource availability and cost (e.g., the availability of catalyst feedstock and electricity cost), and investment costs, all of which will vary by plant. However, the model and the general results presented in this study can still provide a good reference for decision making in technology investment.
One critical implication from this research is that from a cradle-to-gate perspective, fugitive emissions (methane escaped from natural gas production, processing, distribution, and used in the ethylene plants) play a vital role in the realization of expected GHG emissions reductions brought by adopting state-of-the-art or emerging technologies. The authors designed high and low fugitive emission scenarios to investigate the impacts of fugitive emissions. The results showed that the adoption of both state-of-the-art and emerging technologies lead to less greenhouse gas emission reductions at the high fugitive emission estimate than they have at the low fugitive emission estimate. In the high fugitive emissions scenario, fugitive emissions generated during natural gas recovery and processing could counteract or even exceed the reduction of greenhouse gas emissions enabled by state-of-the-art and emerging technologies. The authors made the suggestion that both the U.S. government and the natural gas industry should make intensive efforts to reduce fugitive emissions.
Another major implication of this research is that faster commercialization of emerging technologies can bring more reductions of primary energy consumption (5-38% more cumulative energy savings) and more mitigations of greenhouse gas emissions (7−33% more reduction in the high fugitive emissions scenario and 2−25% more reduction in the low fugitive emissions scenario) in near future, and reduce the risk of diminishing mitigating potentials for greenhouse gas emissions caused by possible high fugitive emissions in the future. This implication is critical for policy makers, investors, manufacturers, and analysts who question whether intensive efforts to bring technology to market faster are worthwhile. These stakeholders can use the results shown in this study to support decision making, to assist further economic analysis, or to enhance strategic planning related to investments in emerging technologies.
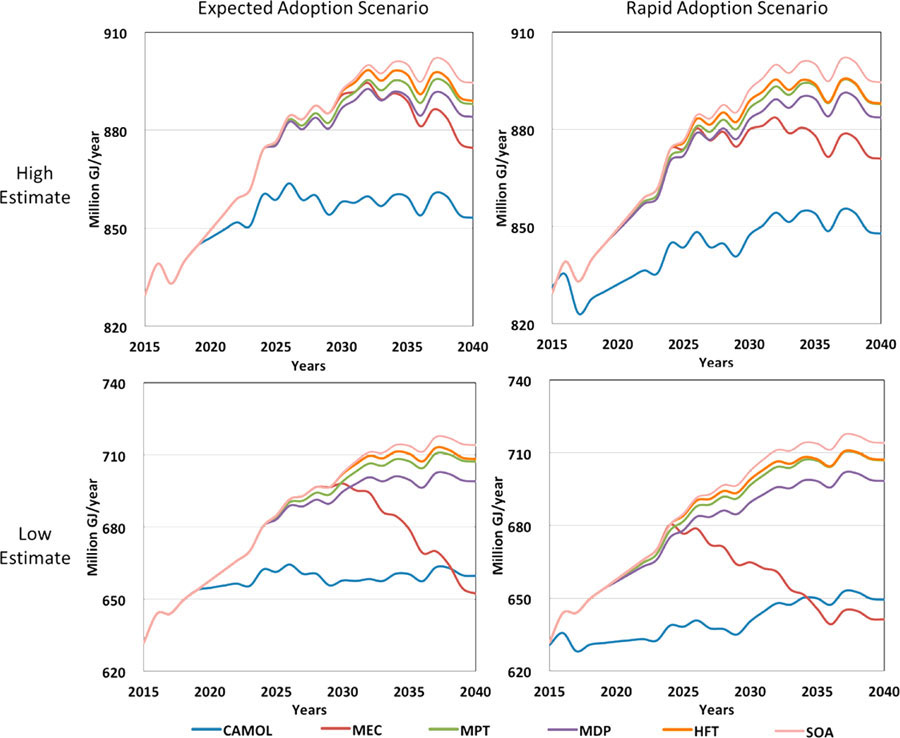
Reproduced with permission from [Y. Yao, D. J. Graziano, M. Riddle, J. Cresko, and E. Masanet, “Prospective energy analysis of emerging technology options for the United States Ethylene Industry ” Industrial & Engineering Chemistry Research 2016, 55 (12), pp 3493–3505. Copyright [2016] American Chemical Society.
Journal Reference
Yuan Yao*1, Diane J. Graziano2, Matthew Riddle3, Joe Cresko4, Eric Masanet*1,5. Prospective Energy Analysis of Emerging Technology Options for the United States Ethylene Industry. Industrial & Chemical Engineering Research, 2016, Volume 55, pp 3493-3505.
[expand title=”Show Affiliations”]- Department of Chemical & Biological Engineering, Northwestern University, 2145 Sheridan Road, Evanston, Illinois 60201, United States.
- Global Security Sciences Division, Argonne National Laboratory, 9700 South Cass Avenue, Argonne, Illinois 60439, United States.
- Energy Systems Division,Argonne National Laboratory, 9700 South Cass Avenue, Argonne, Illinois 60439, United States.
- Advanced Manufacturing Office, United States Department of Energy, 1000 Independence Avenue, SW, Washington, DC 20585, United States.
- Department of Mechanical Engineering, Northwestern University, 2145 Sheridan Road, Evanston, Illinois 60201, United States [/expand]
Go To Ind. Eng. Chem. Res
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.






