Significance Statement
N-heterocyclic carbenes are the most versatile and successful group of organocatalysts developed in recent years. Their numerous benefits include high nucleophilicity and Bronsted-basicity, structural variability, and intriguing chemistry of the carbene reactive center. These benefits have enabled demanding chemical transformations such as addition to double bonds and Umpolung reactions. Polymer chemistry has significantly developed thanks to these advances in organic chemistry. N-Heterocyclic carbene-triggered organopolymerization, for examples can be implemented to synthesize polymers through ring opening polymerization, by condensation reactions and also by conjugate addition.
In most instances, the N-heterocyclic carbenes offer additional benefits, which include polymerization rates and easy control of the end-groups. Unfortunately, the change from laboratory to commercial use, large-scale implementation has been affected by inherent problems that emanate from the use of N-heterocyclic carbenes. These problems include sensitivity to protic compounds, synthetic requirements, and limited storability, all which can be prohibitive for use in the polymer industry.
Researchers led by Professor Michael Buchmeiser at University of Stuttgart in Germany, presented a rapid epoxy curing system with an intention of minimizing the problems stated above while focusing on the benefits which can be derived from using N-heterocyclic carbenes. They presented epoxy resins based on typically used epoxy compounds, carbon-dioxide protected N-heterocyclic carbenes and a number of anhydrides that combine the superior attributes of fast curing at low reaction temperatures, full homogeneity in the form of low viscosity liquids and pot life within the range of hours to days. Their research work is published in European Polymer Journal.
The authors achieved the above attributes through a meticulous tuning of the components. 1,3-Dimethylimidazolium-2-carboxylate as a thermally latent organocatalyst offered facile one-step synthesis and robustness towards atmospheric conditions and high reactivity needed once the polymerization was switched on.
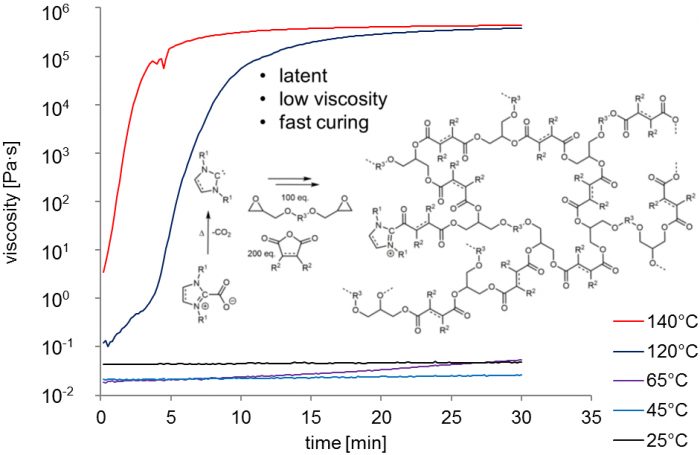
The research team conducted a systematic screening of a number of epoxy compounds and 14 different anhydrides in a bid to identify monomers that were suited for rapid monomer consumption and therefore low curing temperatures. From the study, over 15 different systems were obtained where a homogeneous liquid could be formed in the absence of additional additives.
The authors were able to optimize the curing attributes of the latent one component epoxy compositions implementing thermally triggerable carbon-dioxide protected N-heterocyclic carbenes reference to low viscosity as well as fast curing kinetics at low-temperature conditions. This was possible through the implementation of a well accessible and robust protected N-heterocyclic carbene that was synthesized in a single step from commodity chemicals. The evaluation of commonly used epoxy compounds in combination with an exhaustive range of anhydride components allowed for identification of compositions that could be cured at temperatures of 120 °C or 100 °C.
The mixture provided important pot times of hours to days depending on temperature as well as catalyst loading. A prudent choice of comonomers ensured that fully homogenous, low viscosity and technically sprayable one-component mixtures were obtained, therefore, widening the range of applications. The benefits of N-heterocyclic carbene precursor in combination with the catalytic power of the free N-heterocyclic carbene therefore led to readily applicable systems that made best use of N-heterocyclic carbene organocatalysis, while avoiding the liabilities that normally curtail large-scale application of this form of a catalyst.
Reference
Hagen J. Altmann, Stefan Naumann, Michael R. Buchmeiser. Protected N-heterocyclic carbenes as latent organocatalysts for the low-temperature curing of anhydride-hardened epoxy resins. European Polymer Journal.
Go To European Polymer Journal Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




