Significance
Rational synthesis of materials is highly desirable since the usability of such materials is dependent on the structures and morphology of the resultant materials. For instance, silver nanoparticles have shown numerous potential applications in various fields as a result of their pre-eminent physical and chemical properties resulting from different shapes of particles. As a result, many synthetic approaches have been developed, based on which numerous silver morphologies have been synthesized. Despite such progress in silver synthesis, it is rather unfortunate that the mechanism controlling shape development of silver particles is yet to be fully understood.
Great efforts have since been devoted in a bid to reveal the nature and mechanism responsible for formation of diverse structures. In particular, Chinese Academy of Sciences researchers led by Prof. Yongsheng Han have demonstrated that the interplay of chemical reaction and diffusion is an essential strategy to control the particle shape evolution. Furthermore, it has been established that the control of micro growth environment by chemical diffusion and reaction provides a facile and effective way to shape nanomaterials. Unfortunately, no one has attempted to confirm the role of chemical heterogeneity in the shape evolution of materials. Consequently, a follow up of the morphology variation with the evolution of chemical heterogeneity would serve great in this quest.
Recently, Prof. Han in collaboration with his colleagues conducted a study with the main objective of trying to figure out the role of heterogeneity in relation to the nature and mechanism of formation of diverse structures. Specifically, they intended to provide an alternative means of achieving the same, by controlling the heterogeneity for desired products. Their work is currently published in the research journal, Chemical Engineering & Processing: Process Intensification.
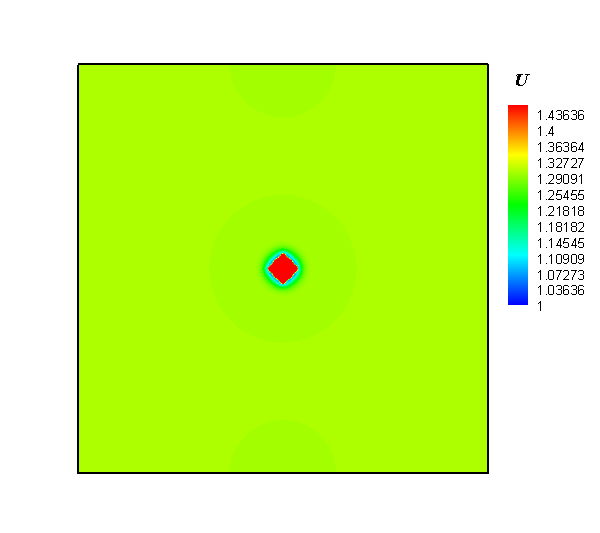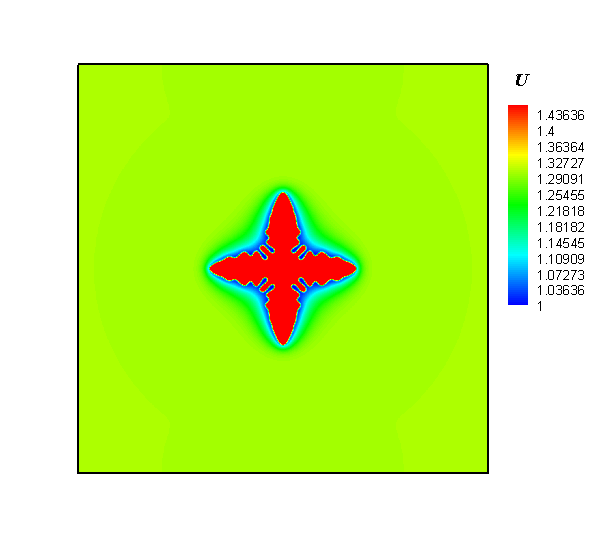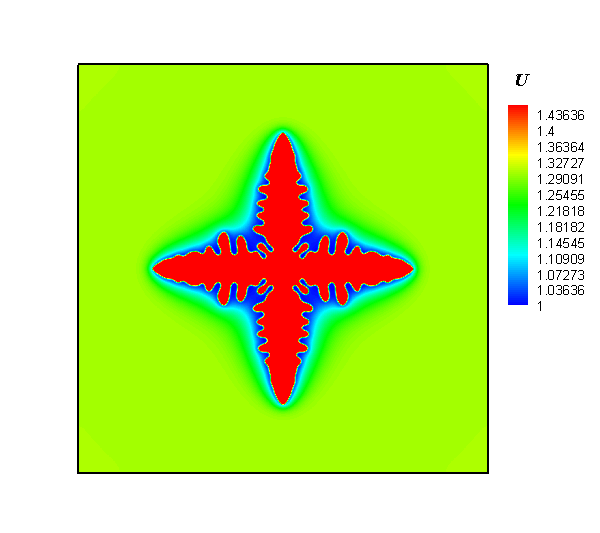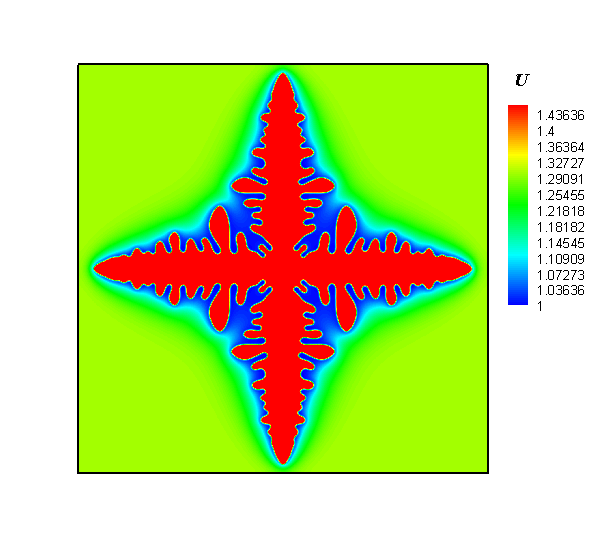
The research team employed a gel system to create a diffusion limitation and to discover how the local concentration heterogeneity influenced the shape development of silver particles in an electrochemical synthesis process. Next, silver dendrites were synthesized around electrode, while polycrystalline spheres were obtained at the forward growth front. Lastly, a temporal evolution of concentration heterogeneity was proposed to interpret the dependence of particle morphology with location.
Interestingly, the authors observed that the shapes of silver particles were dependent on their location. In addition, they observed a mixture of dendritic and spherical particles between the electrode and the growth front. The researchers were also keen to report that by regulating the concentration gradient via alteration of the gel concentration and modification of the reaction rate in electrodeposition, the desired structures were obtained.
In summary, the study successfully demonstrated the synthesis of silver particles via electrochemical deposition in gel solution. Their procedure involved generation of dendrites in the vicinity of an electrode, and the production of polycrystalline spheres in the outer region deviating from the electrode. Altogether, they showed that concentration gradient plays an important role in the shape development of products. As such, control of the local chemical heterogeneity promises a general approach for the rational synthesis of materials.
Reference
Ting Liu, Wei Liu, Yongjun Chen, Tao Yang, Yongsheng Han. Silver morphology indicating the evolution of concentration heterogeneity. Chemical Engineering & Processing: Process Intensification, volume 134 (2018) page 38–44.
Go To Chemical Engineering & Processing: Process Intensification Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



