Significance
Recent technological advances have unveiled the broad applicability of reactive polymers in various fields, including: organic electronics, biotechnology and in medicine. In addition, recently published works have highlighted that the level to which the reactive groups can be addressed provides a facile way to impart stimuli responsiveness/specific selectivity, which is vital for applications that take advantage of the reversible incorporation of guest molecules, stimuli responsive deformations, selective separation of similarly sized biomolecules, or bioinspired self-healing of mechanical and structural properties. As such, microgels based on reactive polymers have become popular with researchers due to their excellent attributes. To date, there has been several published reports describing the preparation of reactive polymeric microgels. Unfortunately, these reports merely highlight the broad interest in developing novel and versatile microgels that can be synthetically tailored and reactively modified to control their structure and properties. Worse off, there is no published work elucidating on the fabrication of reactive microgel using azlactone-containing block copolymers.
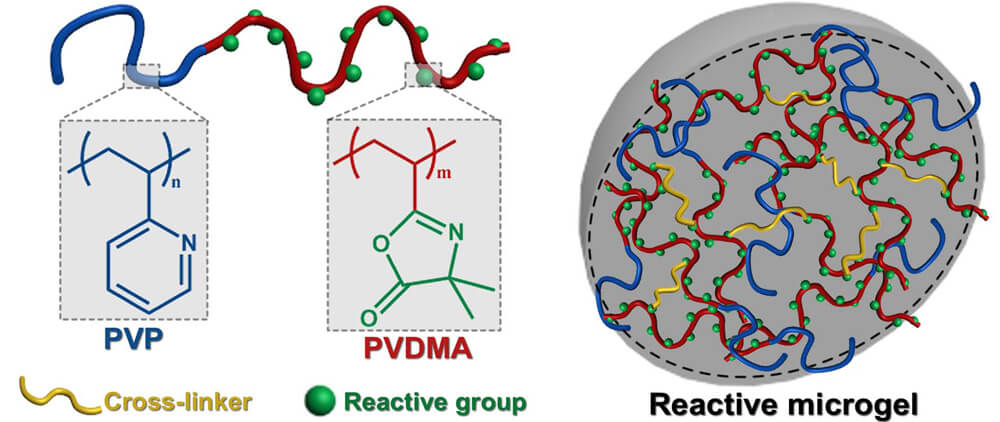
Recently, Professor Xu Wang at Shandong University in collaboration with Dr. Jesse L. Davis, Dr. Bethany M. Aden, and Professor S. Michael Kilbey, II at University of Tennessee and Dr. Bradley S. Lokitz at the Oak Ridge National Laboratory synthesized azlactone-based microgels by successive self-assembly of PVP-b-PVDMA block copolymers in solution. In addition, they modified the resulting nano-assemblies by cross-linking in a selective fashion. Their work is currently published in the research journal, Macromolecules.
The research method employed commenced with the synthesis of PVDMA and PVP-b-PVDMA block copolymers of different molecular weight that were to be used to investigate the self-assembly and guest loading characteristics. Next, the researchers used a diamine/diiodide to cross-link either the PVDMA or the PVP blocks in the self-assembled aggregates. Lastly, a series of functional primary amines were used to investigate the chemical reactivity of the covalently cross-linked PVP-b-PVDMA block copolymers microgels.
The authors observed that the DAB-cross-linked block copolymer microgels swelled in THF, thereby suggesting the formation of a stable, three-dimensional network structure. They also noted that the azlactone-containing block copolymer microgels provided an attractive platform for applications in a wide range of fields, including imaging, catalysis, molecule separation and guest loading for targeted delivery. This was seen to be due to their ability to be reactively modified in ways that allowed their stability or disassembly characteristics to be tailored.
In summary, the Xu Wang and colleagues study presented a facile method involving self-assembly and cross-linking of azlactone-containing block copolymers as an excellent route to fabricate microgels containing reactive “handles” that could be selectively modified to incorporate additional functionality. Generally, in their approach the surfactant-like nature of PVP-b-PVDMA block copolymers was seen to enable the formation of nanoscale aggregates in solvents selective for PVP blocks, and the size of the self-assembled structure could also be manipulated by tailoring block lengths of the block copolymers. Altogether, the advantages derived from the inherent polymerizability of PVDMA and efficacy of reactive modification without formation of byproducts, make azlactone-containing block copolymers useful as building blocks for functional self-assembled systems or soft scaffolds, which in turn make them applicable as nanoreactors or in sensing applications.
PVP-b-PVDMA – poly(2-vinylpyridine)- block-poly(2-vinyl-4,4-dimethylazlactone)
DAB – 1,4-diaminobutane
Reference
Xu Wang, Jesse L. Davis, Bethany M. Aden, Bradley S. Lokitz, and S. Michael Kilbey, II. Versatile Synthesis of Amine-Reactive Microgels by Self-Assembly of Azlactone-Containing Block Copolymers. Macromolecules 2018, volume 51, page 3691−3701.
Go To Macromolecules 2018 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.






