Significance
Per- and polyfluoroalkyl substances (PFAS) have become emblematic of a persistent environmental crisis, not only because of their chemical resilience but also because of their widespread presence in human and ecological systems. Among them, perfluorooctane sulfonic acid (PFOS) is especially troubling. This compound, once heavily used in industrial applications and consumer products, is now recognized as a potent contaminant with direct links to immune suppression, endocrine disruption, and carcinogenic outcomes. The strength of the carbon–fluorine bond that defines PFAS chemistry makes these molecules exceptionally resistant to degradation, allowing them to accumulate in soils, waterways, and biological tissues for decades. What complicates the issue further is the ubiquity of exposure: drinking water has become a critical pathway through which PFAS, at concentrations of just a few parts per trillion, enter human populations. Efforts to mitigate this risk have relied heavily on highly sensitive detection techniques, with liquid chromatography–tandem mass spectrometry (LC-MS/MS) standing as the regulatory gold standard. While robust and precise, this method is not easily deployed at scale. It demands costly infrastructure, highly trained personnel, and labor-intensive sample preparation, effectively restricting its utility to centralized laboratories. The limitations of LC-MS/MS are particularly stark when viewed against the urgent public health imperative: communities require rapid, affordable, and reliable monitoring tools capable of detecting PFAS at concentrations well below the maximum contaminant levels now mandated by national authorities. This gap between regulatory demands and technological readiness has spurred global interest in the development of novel sensing platforms. Fluorescent and electrochemical sensors have drawn attention because of their relative accessibility and design flexibility, yet these technologies often lack the sensitivity or selectivity required for real-world monitoring at environmentally relevant concentrations. More fundamentally, they rarely offer the reversibility and stability necessary for continuous, long-term use in water distribution systems.
Field-effect transistor (FET) sensors present an intriguing alternative. Their electronic readout allows for extremely high sensitivity, and their modular architecture enables the integration of selective capture probes. Despite their success in detecting heavy metals, nutrients, and even biological pathogens, FET-based systems have not previously been applied to PFAS detection. The absence of such efforts reflects both the chemical complexity of PFAS molecules and the challenge of distinguishing them from structurally similar compounds or naturally abundant ions present in tap water.
To this account, Professor Andrew Ferguson, Professor Seth Darling & Professor Junhong Chen from the University of Chicago, the researchers designed a remote-gate FET sensor modified with β-cyclodextrin-functionalized reduced graphene oxide, enabling PFOS detection in tap water with an unprecedented reporting limit of ~250 ppq. By coupling electrical transduction with host–guest recognition chemistry, the device achieved rapid, reversible responses and strong selectivity against most co-contaminants. Mechanistic insights from QCM and molecular simulations revealed the interplay between adsorption dynamics and probe–analyte interactions, guiding strategies for further probe refinement. This work represents the first demonstration of FET-based PFAS detection, establishing a new foundation for real-time, continuous monitoring of persistent contaminants.
The research hinged on the deliberate integration of molecular recognition chemistry with the high sensitivity of transistor-based electronics. The team fabricated remote-gate electrodes by layering reduced graphene oxide onto silicon substrates and functionalizing the surface with β-cyclodextrin molecules via a pyrenebutyric acid linker. This host–guest system was expected to capture PFOS selectively through the hydrophobic cavity of β-CD, while the transistor translated those molecular interactions into measurable electrical signals. Surface characterization confirmed the successful functionalization: water contact angles shifted markedly toward higher hydrophilicity, and XPS spectra revealed chemical features consistent with cyclodextrin attachment.
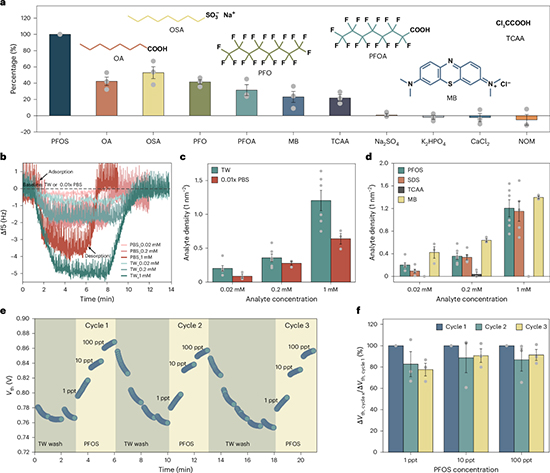
Once the sensing platform was established, the researchers assembled the β-CD-modified electrodes with a commercial MOSFET and Ag/AgCl reference electrode to create the full RGFET system. Initial stabilization cycles were carried out in tap water to equilibrate background PFOS levels before introducing spiked samples. Measurements focused on threshold voltage shifts, which directly reflected changes in surface potential at the water–sensor interface. The results were striking. The device consistently detected PFOS at levels as low as 250 parts per quadrillion—orders of magnitude beneath the maximum contaminant level of 4 ppt. The detection range extended up to 100 ppt, covering the spectrum typically encountered in natural waters. At low concentrations, sensor responses scaled with PFOS levels, while saturation effects at higher concentrations produced nonlinear signal behavior, a well-known phenomenon in FET systems. Importantly, the sensors showed a rapid response time of under two minutes, positioning them as suitable for near-real-time monitoring.
Selectivity tests highlights the practical potential of the system. When exposed to inorganic salts at concentrations thousands of times higher than PFOS, the device exhibited no measurable response. Organic interferents such as methylene blue and trichloroacetic acid produced moderate signals, while structurally related molecules like perfluorooctanoic acid induced partial cross-reactivity. The greatest challenge emerged with sodium dodecyl sulfate, whose surfactant character and structural similarity generated strong interference. This outcome highlighted the need for further probe engineering, yet the strong discrimination achieved against most co-contaminants represented a significant advance.
To probe the mechanisms behind these behaviors, the team employed quartz crystal microbalance assays and molecular dynamics simulations. QCM confirmed that adsorption was necessary but not sufficient for signal generation, with analyte charge properties playing a decisive role. Molecular simulations revealed that PFOS binds tightly within the β-CD cavity, stabilized by van der Waals interactions along its fluorinated backbone, whereas SDS fit less rigidly, allowing alternative configurations that reduced selectivity. These insights not only explained the observed results but also suggested pathways for rational modification of capture probes.
The development of a β-CD functionalized RGFET sensor for PFOS represents more than a technical achievement; it marks a conceptual shift in how persistent water contaminants might be monitored in practice. Existing methods, though precise, remain bound to centralized laboratory infrastructures. By contrast, this work points to a future in which ultra-trace PFAS detection could occur in distributed, low-cost devices embedded directly into municipal or even household systems. Achieving a reporting limit in the parts-per-quadrillion range demonstrates that advanced materials engineering, when paired with molecular recognition chemistry, can rival or even exceed the sensitivity of current gold-standard technologies. Moreover, the reversibility of the sensor response is perhaps its most important feature. Most PFAS detection platforms are either single-use or require elaborate regeneration steps. Here, the ability to restore function by simple rinsing enables a practical route toward long-term deployment. For regulators and utilities, this translates into reduced operational costs and the possibility of continuous water quality monitoring—a transformative shift compared with periodic sampling and delayed laboratory analyses.
The findings also highlights the importance of mechanistic understanding. By integrating QCM experiments with molecular dynamics simulations, the researchers were able to move beyond empirical performance metrics to a nuanced picture of how adsorption, charge interactions, and probe geometry combine to shape sensor behavior. This framework not only validates the observed results but also provides a blueprint for next-generation designs. In particular, the insight that SDS interference arises from flexible binding modes suggests that selective enhancements could be achieved by engineering cyclodextrin rims or incorporating fluorophilic modifications to favor PFAS binding. Such strategies could extend the platform’s applicability across the broader PFAS family, which includes thousands of structurally diverse molecules. The broader implications extend into public health, environmental governance, and technology transfer. Communities grappling with PFAS contamination often lack timely data, which fuels mistrust and hampers mitigation efforts. Deployable sensors like the RGFET platform could democratize access to water quality information, empowering citizens and providing regulators with the high-resolution data needed to enforce safety standards. From a commercialization perspective, the modular design of the sensor—relying on inexpensive transistors and scalable graphene derivatives—offers a viable path to manufacturing. When integrated with wireless communication systems, such devices could form part of a distributed sensor network, providing continuous surveillance of water infrastructure.
Reference
Wang, Y., Jang, HJ., Topel, M. et al. Reversible parts-per-trillion-level detection of perfluorooctane sulfonic acid in tap water using field-effect transistor sensors. Nat Water (2025). https://doi.org/10.1038/s44221-025-00505-9
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



