Significance
Photopolymerization, a process that uses light to initiate polymerization reactions, is a cornerstone in the creation of complex polymer networks. It is pivotal in applications ranging from 3D printing to the fabrication of microelectronics and photoadhesives. Conversely, photoprocessing, which involves the light-induced cleavage of polymer networks, is crucial for applications like photoresists and drug delivery systems. Historically, the simultaneous application of these two technologies on a single material has been hampered by their inherent incompatibility; photopolymerization necessitates the stability of the material under light, whereas photoprocessing relies on the material’s ability to break down under similar conditions.
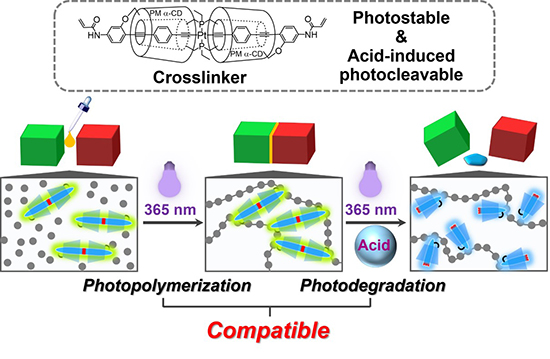
A new study published in Angewandte Chemie International Edition and led by Takashi Kaneko, Dr. Go Russell, Yutaro Kawano, Prof. Dr. Hiroshi Masai, Prof. Dr. Jun Terao from the University of Tokyo, the authors developed a new methodology for fabrication of photoprocessable materials using an acid-induced photocleavable platinum–acetylide crosslinker that has the dual functionality of photopolymerization and photoprocessing within the same material system under the influence of UV light and acidic conditions. This innovation not only opens new avenues for the manipulation and functionalization of polymer materials but also addresses the longstanding challenge of integrating photopolymerization and photoprocessing technologies. Moreover, the findings from these experiments not only confirmed the feasibility of this innovative approach but also provided insights into the underlying mechanisms and potential applications.
The initial set of experiments focused on synthesizing the platinum–acetylide complex, which is central to the dual functionality of the material. This complex was designed to be stable under neutral conditions, allowing for photopolymerization, but susceptible to photocleavage in acidic environments, enabling photoprocessing. The researchers characterized the synthesized complex using various spectroscopic techniques, including NMR, UV-Vis, and fluorescence spectroscopy, to confirm its structure and photoreactivity and demonstrated the synthesized platinum–acetylide complex has the desired stability under neutral conditions and reactivity under acidic conditions.
Afterward, the authors conducted photopolymerization experiments using the synthesized platinum–acetylide complex as a crosslinker. They mixed the complex with monomers and a photoinitiator, exposing the mixture to 365 nm UV light to initiate polymerization. The formation of polymer networks was monitored using rheological measurements, tracking the evolution of the material’s viscoelastic properties. The results demonstrated the material’s ability to undergo photopolymerization, forming stable polymer networks.
To test the photoprocessing capability, the University of Tokyo team exposed the polymerized material to acidic conditions followed by irradiation with the same 365 nm UV light. They used HCl as the acidic additive and observed the material’s response. The acid-induced photocleavage of the platinum–acetylide crosslinker was expected to lead to the softening or degradation of the polymer networks. The extent of degradation was assessed through changes in the material’s mass, rheological properties, and luminescence, indicating successful photoprocessing under the combined influence of acid and UV light. Additionally, the researchers explored its application in creating photoadhesives that can be attached and detached on-demand. They also prepared a photoadhesive by photopolymerizing the material between two substrates. The adhesion strength was measured to assess the effectiveness of the photopolymerization process. Subsequently, the adhesive bond was exposed to acidic conditions and UV light to induce detachment. The ease and efficiency of the detachment process were evaluated, showcasing the material’s potential as a reversible adhesive system.
The team also investigated the possibility of using visible light for photoprocessing because UV light has some limitations in certain applications. They introduced a photosensitizer that could facilitate the acid-induced photocleavage of the platinum–acetylide crosslinker under visible light. The experiments involved irradiating the material with 470 nm visible light in the presence of the photosensitizer and acidic conditions, observing the effects on the material’s properties to confirm the viability of visible light-induced photoprocessing. Indeed, the introduction of a photosensitizer expanded the material’s functionality to visible light, opening up broader applications beyond the limitations of UV light.
According to the authors, by introducing the acid-induced photocleavable crosslinker, based on a platinum–acetylide complex, they successfully enabled the dual functionality of photopolymerization and photoprocessing under the same light conditions (365 nm UV light). The key to this dual functionality lies in the selective activation of the photocleavable crosslinker in the presence of an acidic additive. In neutral conditions, photopolymerization proceeds unimpeded, forming robust polymer networks. However, when exposed to an acidic environment, the same polymer networks become susceptible to photodegradation under the same light conditions. This selective activation is made possible by the unique properties of the platinum–acetylide complex, which remains stable until exposed to the acidic conditions that trigger its photocleavage. In conclusion, the work of Professor Jun Terao and colleagues showcases the innovative potential of combining photopolymerization and photoprocessing technologies in a single material through the use of acid-induced photocleavable crosslinkers, offering new possibilities for the design and application of photoprocessable materials in various industries ranging from electronics to biomedical engineering.
Reference
Kaneko T, Russell GM, Kawano Y, Masai H, Terao J. Fabrication of Photoprocessable Materials via Photopolymerization Using an Acid-Induced Photocleavable Platinum-Acetylide Crosslinker. Angew Chem Int Ed Engl. 2023;62(26):e202305374. doi: 10.1002/anie.202305374.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



