Significance
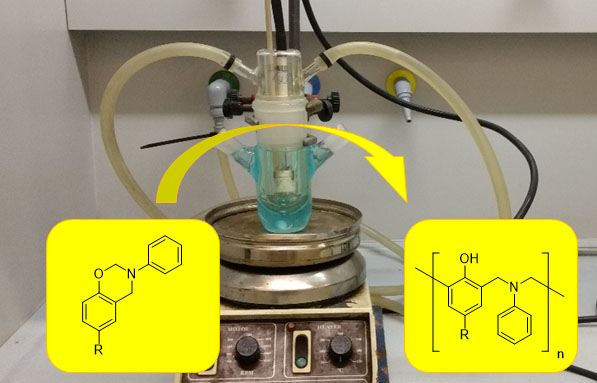
Polybenzoxazines, a type of phenolic resins, come from the polymerization of 1,3-benzoxazine monomers whose preparation is easy and cheap, starting from commercially available phenols and amines. These materials possess many attractive attributes such as low water absorption, near-zero volume changes during polymerization, very high chemical resistance, high char yield among other interesting properties. Nonetheless, the fact that polymerization takes place at rather high temperatures is one of their main drawbacks, not only by the energetic expenditure required, but also because monomeric 1,3-benzoxazines undergo decomposition and hydrolysis at high temperature. Recent studies have shown that catalyst addition can modestly reduce the polymerization temperature. This has in turn led to the wide spread utilization of catalysts to promote ring-opening polymerization. Regardless, so far no one has attempted room temperature synthesis of polybenzoxazines starting from monofunctional benzoxazines without the help of any catalyst or initiator. In addition, no record of any attempt to synthesize polybenzoxazines in water exists. Therefore, it would be very advantageous to find a sustainable method to polymerize mono-1,3-benzoxazines in aqueous solutions at room temperature.
Recently, Universitat Autònoma de Barcelona researchers: Dr. Jordi Salabert a Post-doctoral researcher, Associate professor Dr. Rosa M. Sebastián and Professor Dr. Jordi Marquet tested if the direct ring-opening polymerization of benzoxazines could be elicited photochemically at room temperature, taking advantage of the topologically controlled Coulombic interactions that would be produced in the benzoxazine structure upon UV irradiation. They also were the first to undertake the same using water at ambient room temperature. Their work is currently published in the research journal, Macromolecules.
Briefly, the research method employed consists in the preparation of solutions of 1,3-benzoxazines and their irradiation, using appropriate lamps and immersion reactiors. Firstly, water/organic solvent mixtures were tested, then, in a more sustainable approach, water media using a surfactant agent was performed. Benzoxazine monomers were previously prepared following two well-known methodologies from phenols, paraformaldehyde and triazine derivative in bulk or in the presence of an organic solvent, 1,4-dioxane. Monomers and obtained polymers were characterize using conventional techniques, such as nuclear magnetic resonance. Molecular weights and polydispersities of polymers were obtained by gel permeation chromatography.
The authors observed that irradiation of ‘electron-withdrawing groups’-substituted benzoxazines produced opening of the oxazine ring as expected, but no propagation of the polymerization was seen probably due to the low nucleophilicity of the intermediate phenolic species. However, with more nucleophilic electron-donor substituted benzoxazines the photopolymerization proceeded smoothly, with excellent conversions and modest degrees of polymerization. The researchers also noted that the produced materials showed a similar degree of polymerization to the one observed in the non-cross-linked soluble fraction when monobenzoxazines were polymerized under thermal conditions but having a lower dispersity.
In summary, Universitat Autònoma de Barcelona scientists demonstrated that by taking advantage of the topology of the structure of N-phenyl mono- 1,3- benzoxazines and the fact that they absorb in the UV range, it was possible to photochemically induce the O−alkyl bond cleavage and thus the ring opening through an intramolecular electron transfer process. Altogether, the model substituted monobenzoxazines could be photochemically polymerized in aqueous solutions at room temperature.
Reference
Jordi Salabert, Rosa María Sebastián, Jordi Marquet. Photochemical Polymerization of N-Phenyl Mono-1,3-benzoxazines in Aqueous Media . Macromolecules 2018, volume 51, page 3672−3679.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.




